Dr. Aditi Mehta, M18558, Dr. Bhavna Chawla, Dr. Neelam Pushker, Dr. Mandeep Singh Bajaj
![]()
Introduction
Capillary haemangiomas are one of the most common benign tumours of infants, presenting within the first year of life with an estimated prevalence in Indian studies of 0.1 to 0.28%(1) About 80% of the haemangiomas are located in the head and neck region.(2) The periocular lesions can range from strawberry nevi to deeper subcutaneous bluish masses with or without post septal extension.A history of prematurity, low birth-weight, placental anomalies and multiple pregnancies is shown to have a higher incidence of infantile haemangiomas.(3, 4)They are usually noted at birth, exhibit rapid postnatal growth followed by slow involution, often leading to a complete regression.Most haemangiomas can be diagnosed on clinical examination.Imaging with ultrasonography or magnetic resonance imaging is helpful to document vessel density, volumetric size and delineate postseptal extent of deep orbital haemangiomas.Regression rate in periorbital capillary haemangiomas has been studied to be 30% by age three and70% by age seven.(5, 6) The growth patterns of superficial (strawberry nevi) and deeper haemangiomas (orbital and subcutaneous) differ as the latter have a later onset and prolonged period of growth compared to strawberry nevi.(7)
Associated ophthalmic complications include mechanical ptosis, astigmatism and anisometropic amblyopia, deprivational amblyopia (visual axis obstruction), disfiguring proptosis, exposure keratopathy, optic nerve compressionor lesion necrosis or infection.(7-9)Periocular capillary haemangiomas may occur simultaneously with oral, nasal, subglottic, and paratracheal haemangiomas which may lead to respiratory obstruction and asphyxia.
In absence of these complications, the treatment for capillary haemangiomas is limited to observation for most cases because of the natural course of spontaneous regression. For the complicated cases, various established treatment modalities include medical therapy with glucocorticoids, systemic beta-blockers, immunomodulators, laser ablation, radiation, embolisation or surgical excision. A review by Léauté-Labrèze published in 2017 recommended that most haemangioma lesions did not require therapy. When treatment was indicated oral propranolol was the first line of choice and a minimum period of 6 months of treatment was necessary to give good results.(10)
Review of Literature:
Various studies on capillary haemangiomas have discussed their presentation, clinical course, diagnostic modalities and treatment options. The role of beta blockers for treating capillary haemangiomas was serendipitously discovered by Léauté-Labrèze in 2008 when they observed a dramatic decrease in size of cutaneous capillary haemangiomas in an infant being treated with propranolol for a cardiac indication.(11)
The proposed mechanisms of action for beta blockers include vasoconstriction, suppression of angiogensis through the hypoxia-inducible factor 1a vascular endothelial growth factor (HIF-1a- VEGF) pathway and induction of apoptosis of the endothelial cells.(12)In addition, propranolol has shown to have inhibitory action on microRNAs responsible for stem cell like property of the endothelial cells lining the haemangioma. While the exact mechanism is unknown, it is likely that a combination of the above effects is responsible for the therapeutic action of beta blockers.(13-15)
Ames et al evaluated the current trends in the medical management of infantile haemangiomas and concluded that since 2008, propranolol has become the first-line therapy, whereas other medical treatments are used less frequently or when propranolol is unsuccessful.(16) Oral propranolol hydrochloride has obtained FDA approval in March 2014 for treatment of proliferating infantile haemangiomas requiring systemic therapy.(17)
These drugs can be administered via systemic (oral), topical and intralesional routes.(18, 19) A study done by Hao et al in 2011 in rabbit eyes evaluated the distribution of propranolol in periocular tissues after oral, intravenous and topical administration. They concluded that topical administration can provide increased concentrations of propranolol in the periocular tissues and is superior to systemic administration for the treatment of periocular haemangiomas.(20) Awadein et al comparatively described the use of intralesional propranolol injection and intralesional triamcinolone in periocular capillary haemangiomas. They documented a statistically comparable reduction in the size of haemangioma, astigmatic error, and degree of ptosis in both groups. No adverse effects were reported during or after intralesional propranolol injection.(21)
A randomized control trial of 45 patients by Zaher et al compared the efficacy and safety of topical and intralesional propranolol with oral propranolol. Excellent response (complete resolution) was achieved in 9/15 patients on 2mg/kg/day oral propranolol (60%), 3/15 in patients on topical propranolol 1% ointment twice daily (20%) and 2/15 in weekly 1mg/ml intralesional propranolol (13.3%), (p value: 0.04). As regard to safety, all 3 modalities proved safe with no major side effects. They concluded that further research is needed to help establish clear guidelines and reach best formulations. Topically administered propranolol could be considered in patients at risk of potential side effects from oral administration. However, as intralesional application did not offer any more benefits, it could not be recommended.(19)A pilot study by Torres-Pradilla et al in 2013 discussed the role of intralesional propranolol in infantile haemangiomas in six patients (ages between 2 to 12 months) with focal (superficial or mixed) lesions that were of cosmetic concern. All patients received one injection of intralesional propranolol 1mg/ml concentration in a dose of 0.2ml/cm, maximum volume of 1 ml. The clinical response was noted at 4 weeks and injection was repeated in 5 patients who showed some response in terms of reduction in size. Only one patient who showed some response (colour change) with the second dose received the third dose. None of the patients showed any systemic side effects. They concluded that overall beneficial role of intralesional propranolol in infantile haemangiomas was guarded. They attributed the failure of intralesional propranolol in their study to erratic absorption, lack of a local deposit after intralesional injection, inappropriately low dose and number of injections and a higher mean age of patients in their study (7.3 months).(22)
Side effects of systemic propranolol have been evaluated in many studies. In a large systematic review of IHs treated with propranolol, Marqueling et al. reported the most common adverse events as changes in sleep (11.4%) and acrocyanosis (5.1%). The incidence of serious adverse events was rare (symptomatic hypotension in 0.4%, hypoglycaemia in 0.3%, and symptomatic bradycardia in 0.08% patients).(23) Likewise, restless sleep, constipation, and cold extremities were also observed by de Graaf et al. but they concluded that side effects such as symptomatic hypoglycaemia, hypotension, and bronchial hyper-reactivity that needed intervention and/or close monitoring were infrequent and not dose-dependent.(24)
Holland et al reviewed a case series of 21 patients who developed hypoglycaemia related to propranolol use.(25) Propranolol is thought to cause hypoglycaemia by inhibiting glycogenolysis, gluconeogenesis and lipolysis, in a non dose dependent mechanism. In addition, beta blockers mask the early sympathetic signs of hypoglycaemia (tachycardia, sweating and palpitations). It is therefore recommended that in patients with reduced calorie intake due to inter-current illness, propranolol should be temporarily discontinued.(26) It is recommended that those with large haemangiomas should be referred to a paediatric cardiologist before commencing propranolol.(27,28)
Regarding intralesional injections, limited literature is available. The most serious and feared complication is central retinal artery occlusion (CRAO).(29)A paediatric dermatology research workshop in 2005 cautioned regarding the rare risk of inadvertent intravascular/ophthalmic artery embolization with permanent visual loss during administration of intralesional steroids.(30) Bang et al recommended that while administering intralesional corticosteroid, retinal vessels should be examined during and after injection to monitor for central retinal artery occlusion.(18) Though this has been documented with steroid injection, it may be a potential risk with intralesional propranolol also. In their study comparing intralesional propranolol with intralesional triamcinolone, Awadein et al did not report any adverse effects during or after intralesional propranolol injection.(21)
To summarise, oral propranolol is the established first line treatment for infantile haemangiomas. Though rare, systemic side effects can be associated with oral propranolol. Direct drug delivery with possible additional beneficial injection procedure action with intralesional propranolol needs to be investigated. We planned to evaluate intralesional as an alternative to oral and compare efficacy as well as side effects and safety profile. The primary outcome parameters were reduction in surface area of lesion. The change in appearance, color and ptosis and the keratometric astigmatism were also assessed. Patients were monitored for side effects.
Materials and Methods
Study Design: Randomized Phase II Clinical Trial
Duration of study: One and a half years
Sample Size: Twenty patients
The trial was approved by the Institute ethics committee of the place of study(Reference number: IESC/T-446/26.08.2015, RT-8/27.11.2015) and has been registered under theClinical Trial Registry of India (CTRI; Registration Number: CTRI/2017/08/009440).
Patient Selection: Twenty consecutive newly diagnosed, treatment naïve cases of periorbital and eyelid capillary haemangiomas attending the centre were recruited. They were randomized to two groups using a computer generated random number table. The groups were not matched for age or gender. There was no control “observation only” group.
The inclusion and exclusion criteria are enlisted as follows:
Inclusion Criteria:
Newly diagnosed treatment naïve cases of periocular and eyelid capillary haemangioma whose guardians signed a written informed consent were recruited.
Exclusion Criteria:
Patients having intraorbital spread of lesion, complicated lesions with ulceration and necrosis, or previously treated patients were excluded. Patients with a history of systemic illnesses including recurrent breathing problem/ asthma; cardiac problems including AV block, CHF, sinus bradycardia, seizures or developmental delay; abnormal finidngs on blood investigations or a failure to obtain cardiology clearance for intervention were excluded from the study. The patients whose guardians didn’t sign the consent or were not willing for investigations, treatment or follow up were also excluded.
Baseline evaluation:
Clinical history included age of onset and progression of lesion if any. Birth history and details of gestation were also noted. Systemic evaluation including general physical examination, cardiovascular, respiratory system and per abdomen examination was done. Pre –interventional investigations at the time of enrolment included clinical documentation of size, colour and appearance of lesion. Any associated complications like ptosis, strabismus or other changes were recorded. The fixation preference and dilated retinoscopy of all patients was performed prior to intervention. Visual acuity was also recorded whenever possible. The corneal astigmatism was recorded using a hand held auto refractometer (Retinomax 3+ (Rmax); Nikon Inc., Japan).
Imaging of the lesions was done using orbital ultrasonography and magnetic resonance imaging (MRI) scan of head and orbit. Abdominal ultrasonography, when indicated, was done to document any visceral haemangiomas. Preoperative haemoglobin, bleeding time, clotting time, random blood sugar, liver and kidney function tests were tested when required.
Photographs of the lesions were taken at baseline and on subsequent follow up.
Study Groups:
1) Study Group A: 10 patients received oral propranolol in a dose of 1mg/kg/day in two divided doses on day 1 & 2; then 2mg/kg/day in two divided doses on day 3 & 4 and then 3mg/kg/day in two divided doses, continued till six months. At the end of six months, the dose was tapered over a period of 6 days and then stopped. Final evaluation was done at 6 months.
2) Study Group B: 10 patients received intralesional propranolol in the form of a single injection with a 26 gauge needle. The propranolol used was prepared in strength of 1mg/ml concentration and was provided by the hospital pharmacy of the place of study. The dose for intralesional propranolol was 0.2 ml (of 1mg/ml concentration) per cm of the greatest linear dimension (GLD) of lesion with the maximum injected dose being 1ml. Injection was administered under general anaesthesia in the operation theatre with monitoring of heart rate and blood pressure. All patients received three injections – at baseline, between 4 to 6 weeks and between 8 to 12 weeks. Follow up was continued and final evaluation was done at 6 months.
The two groups of patients were admitted and underwent cardiovascular & respiratory monitoring for the first 48 hours after treatment. Heart rate, blood pressure and respiratory system were also examined on subsequent follow up visits done at week 1, 2, 4, 6, 8, 12 and then at 4 months and 6 months.
Observation Parameters
Efficacy of therapy was measured as
- Regression in size on clinical examination:
- Percentage decrease in area (length along longest linear dimension and width perpendicular to the longest linear dimension; measured clinically using digital callipers)
- Grading for reduction in area on the basis of the following scale (modified and adapted from the study done by Awadein et al).(21)
| Excellent | >90% |
| Very good | 70-89.9% |
| Good | 50-69.9% |
| Fair | 30-49.9% |
| Poor | <30% |
Table 1: Grading scale for percentage area reduction
- Reduction in degree of ptosis
- Mild Ptosis: <2mm
- Moderate Ptosis: 2-4mm
- Severe Ptosis: >4mm
- Regression in colour (from dark red to pale pink)
- Regression in appearance (from elevated to flat)
- Change in astigmatism
- Side effects: Systemic side effects including cold extremities, change in feeding habits, constipation, gastric reflux or regurgitation, history of jitteriness or lethargy were noted. Other symptoms evaluated were wheezing, bradycardia or hypoglycaemia. Local side effects like necrosis or ulceration at lesion site were documented. Any other side effects as reported by guardians or noted on evaluation during follow up were also noted.
Analysis of Study
Data was recorded in a predesignedproforma and Excel spreadsheet. Categorical variables were summarised as frequencies. Quantitative variables were summarised as mean ± SD or median if non-normally distributed. Quantitative data results were compared and interpreted using the two-sided students t test with an alpha error of 5% and a power of 80%. A p value of ≤ 0.05 was considered statistically significant.
Results
Baseline Characteristics in groups 1 and 2
Twenty patients were recruited after taking written informed consent from the guardians. They were divided into 2 groups on the basis of random number tables with 10 patients in group 1 (oral propranolol, case 1 to case 10) and 10 patients in group 2 (intralesional propranolol, case 11 to case 20). The baseline characteristics were analysed and were statistically comparable except for gender. These are summarized below.
| Demographics | Group 1 | Group 2 | p value |
| Age (median) | 8 months | 10 months | 0.107 |
| Gender | 7 female, 3 male | 2 female, 8 male | 0.02 |
| Birth complications | 0.785 | ||
| Uncomplicated
Breech Twins Low birth weight |
8
1 1 0 |
7
1 1 1 |
|
| Period of gestation | 0.531 | ||
| Term
Preterm |
9
1 |
8
2 |
|
| Type of birth | 0.606 | ||
| Normal vaginal delivery
Caesarean Section |
8
2 |
7
3 |
|
| Type of lesion | 0.475 | ||
| Skin surface localized
Skin surface extensive Conjunctival localized Deep extensive |
1
1 3 5 |
4
1 2 3 |
|
| Area (median) | 331.53 mm2 | 257.62 mm2 | 0.205 |
| Colour | 0.549 | ||
| Dark red-blue
Red Pink Same as surrounding skin |
7
2 1 0 |
7
3 0 0 |
|
| Appearance | 0.08 | ||
| Elevated
Mildly elevated Flat |
7
3 0 |
9
1 0 |
|
| Ptosis | 0.98 | ||
| None
Mild<2mm Moderate 2-4mm Severe>4mm |
4
0 1 2 |
2
1 4 3 |
|
| Astigmatism | 2.8588 D at 179.8 degrees | 1.617 D at 90.16 degrees | 0.502 |
Table 2: Summary of baseline characteristics in groups 1 and 2
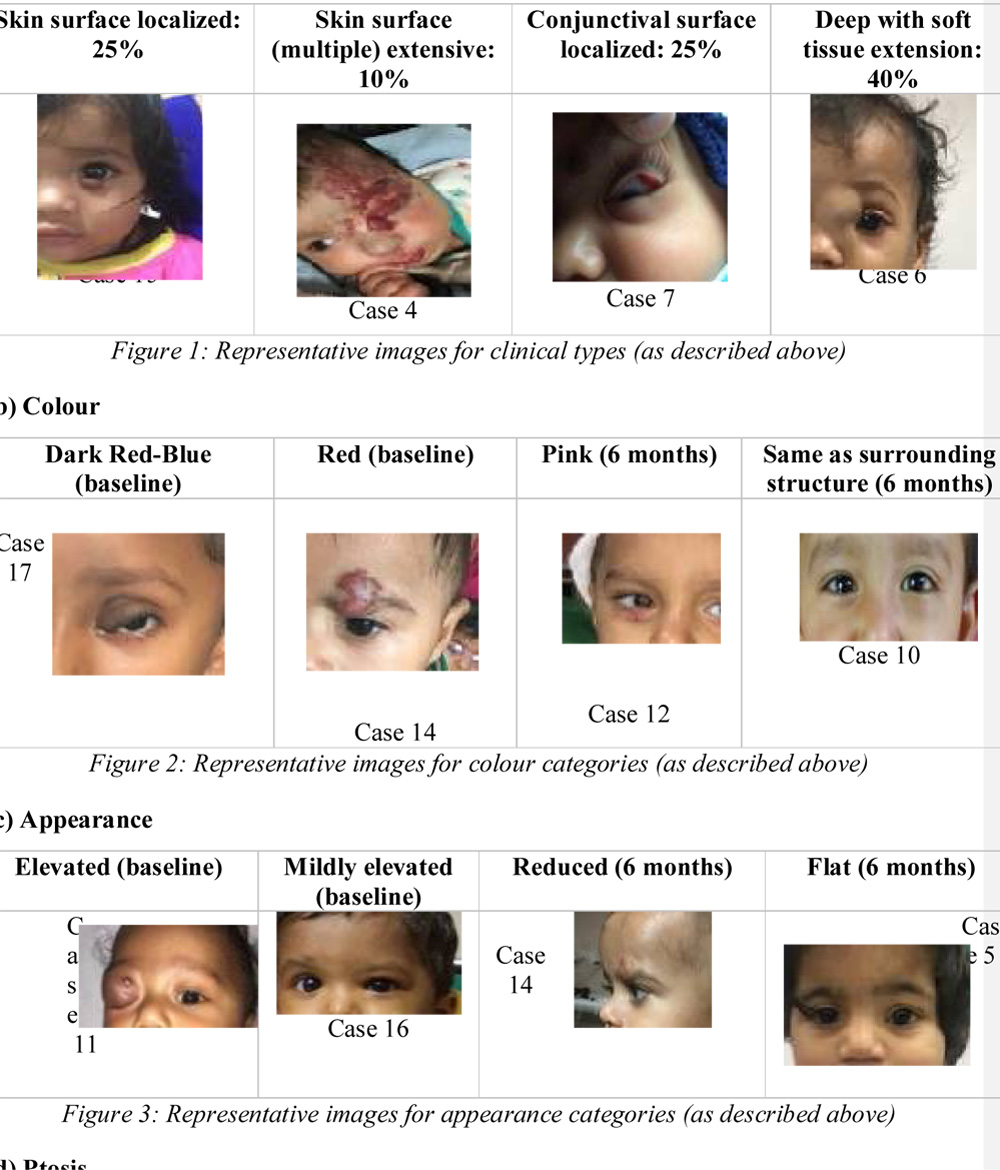
Representative images
a) Type of lesion
d) Ptosis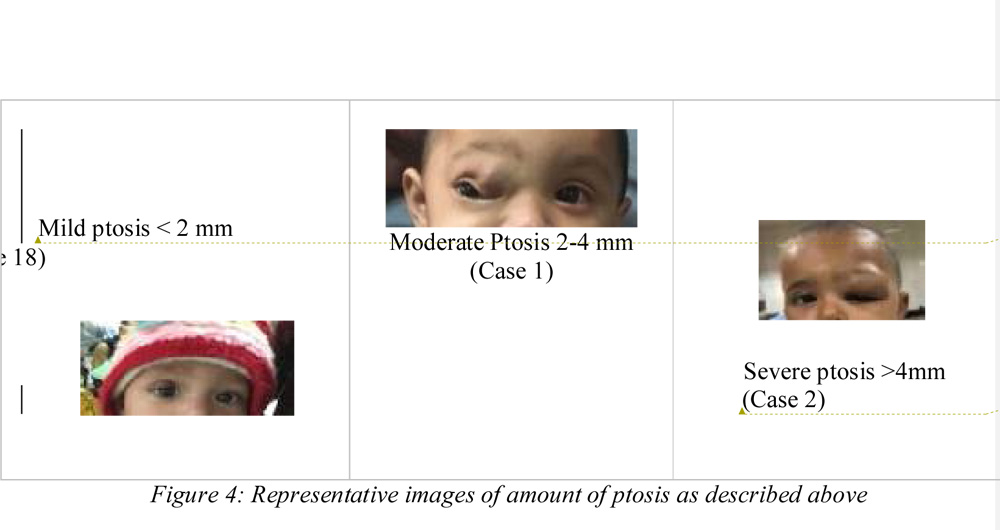
Outcome:
- a) Change in area of lesion:
All ten patients in the group 1 (oral propranolol group) showed a significant response to treatment. The average percentage area reduction was 83.48% ±11.67% at the end of six months. (p value 0.0019). A similar response was seen in all ten patients in group 2 who received 3 intralesional injections of propranolol at 4-6 week intervals. At 6 months, the average percentage area reduction was 67.78% ± 21.71% (p value 0.0019).
| Group 1 n=10 | Group 2 n=10 | |
| Size of lesion (area) at baseline (mean +/- SD) | 615.77 +/- 592.67 mm2 | 332.3 +/- 261.94 mm2 |
| Size of lesion (area) at 6 months (mean +/- SD) | 130.34 +/- 162.86 mm2 | 132.89 +/- 149.76 mm2 |
| Average percentage area reduction (mean +/- SD) | 83.48% +/- 11.67 | 67.78% +/- 21.71 |
| p value | 0.0019 | 0.0019 |
| p value group 1 vs. group 2 | 0.056 (t test) | |
Table 3: Change in area of lesion from baseline to 6 months in groups 1 and 2
The patients in group 1 who received oral propranolol showed an overall greater percentage reduction in area with treatment as compared to the patients in group 2 who received the intralesional injection. This difference in percentage reduction in area was significant by statistical analysis at 4 months (p value 0.047). However, this difference was not noted at the 6 month follow up (p value 0.056). Thus, intralesional propranolol seemed comparable to oral propranolol therapy at the 6 month analysis.
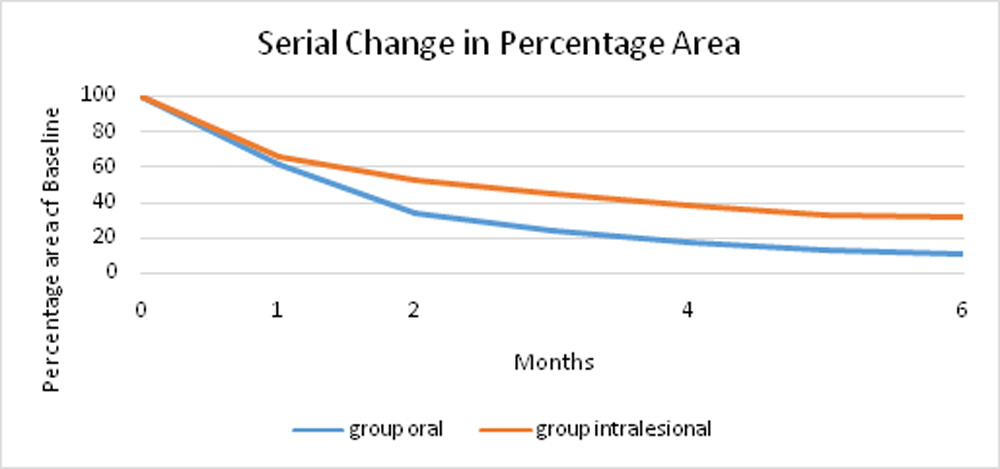
Figure 5: Serial reduction in percentage area in groups 1 & 2
The percentage reduction values were further divided into categories in order to grade the response as excellent, very good, good, fair and poor.
| Grade | Reduction in area% at 6 months | Group 1 | Group 2 |
| Excellent | >90% | 4 | 3 |
| Very good | 70-89% | 4 | 2 |
| Good | 50-69% | 2 | 3 |
| Fair | 30-49% | 0 | 2 |
| Poor | <30% | 0 | 0 |
| p value (Fishers exact test) | 0.56 | ||
Table 4: Percentage reduction in area size according to grading scale in groups 1 & 2
Forty per cent of the patients in group 1 showed an excellent response as compared to 30% in group 2. All ten patients in group 1 had more than 65% improvement in area and the response was good or higher. As compared to this, only 80% of the patients in group 2 had a good or higher response. Twenty per cent patients in group 2 had a fair response with the least responsive patient showing only 31.48% reduction in area. Thus, even though there was a better response in group 1 as compared to group 2, this was not significant statistically. (p value 0.56). The average area at baseline and on follow up at 1,2,4 and 6 months in each group is summarised in table 5.
| Time point | Group 1 Average area in mm2 | Group 2 Average area in mm2 |
| Baseline | 615.77 | 332.3 |
| 1 month | 415.2 | 251.12 |
| 2 months | 276.75 | 202.12 |
| 4 months | 160.15 | 151.5 |
| 6 months | 130.34 | 132.89 |
Table 5: Summary of Area at baseline and on follow up
b) Astigmatism
- The keratometry was done with the help of hand held auto kerato-refractometer and the amount of corneal astigmatism in the affected eye was noted at baseline and at six months. The change in astigmatism after treatment in each group was compared as vectors.(31) This was done by calculating the mean preoperative and postoperative centroids, as described by Holladay et al.(32) Briefly, polar values were converted to Cartesian values using the following equations:x = cylinder × cos (2 × axis) andy = cylinder × sin (2 × axis)In the formulas, the angle of the axis of astigmatism is doubled to give the correct x and y values. The centroid, or mean of a set of x and y values, was calculated by independently finding the mean of each variable (xm, ym). The centroid (mean astigmatism) was then converted back to standard polar notation as follows:Cylinder = (xm2 + ym2)0:5 and Angle = 1/2 arc tan (ym / xm )(If xm and ym > 0, then axis = angle; if xm < 0, then axis = angle + 90 degrees; if xm > 0 and ym < 0, then axis = angle + 180 degrees).The table represents the amount of astigmatism in dioptres at the steep axis, along with the axis at baseline and at 6 months in all 20 patients.
| Baseline (amount in D) | Baseline (axis in degrees) | 6 months (amount in D) | 6 months (axis in degrees) | p value | |
| Group 1 | 2.86 | 179.8 | 1.13 | 179.8 | 0.0045 |
| Group 2 | 1.62 | 90.2 | 0.75 | 179.9 | 0.0001 |
Table 6: Astigmatism (steep axis Dioptre and degrees) at baseline and 6 months
The astigmatism analysis was done using the method described by Holladay, detailed above. Both groups showed a significant amount of reduction in mean dioptres of astigmatism. There was a 60.53% reduction in amount of astigmatism ingroup 1(p value 0.0045) and a 53.54% reduction in amount of astigmatism in group 2 (p value 0.0001). On comparing the amount of reduction in dioptres of astigmatism between the two groups, there was no significant difference; p value 0.49.
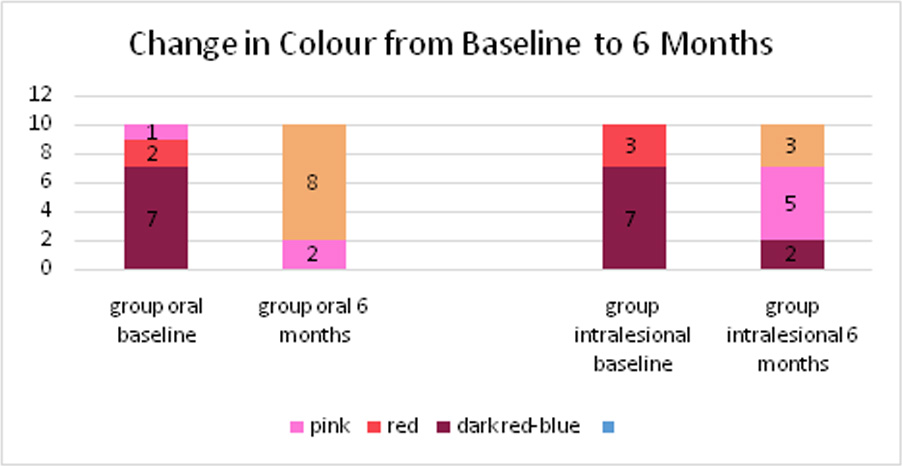
c) Change in colour:
In group 1, 70% had a dark red-blue colour at baseline. At the end of 6 months, 80% had colour same as surrounding structure and 20% had a pink appearing lesion showing a significant improvement (p value 0.0404).
In group 2, 70% patients had a dark red-blue colour at baseline and 30% had red appearing lesions. At the end of 6 months, 20% patients remained to have a dark red-blue colour and 50% had a pink appearing lesion. Only 30% patients had a colour that was same as the surrounding skin. This change in colour was statistically significant within in group 2 (p value 0.02). The overall improvement in colour was better in group 1 than group 2 with 80% patients having colour same as surrounding structure at the end of 6 months, compared to only 30% in group 2 having the colour same as surrounding structure.
 d) Change in Appearance:
d) Change in Appearance:
- The change in appearance in terms of elevation of lesion was also documented. Appearance categories were defined subjectively as grade 2 if significantly elevated, mild elevation/ reduction from baseline but not flat as grade 1 and flat as grade 0. At baseline, 70% patients in group 1 had lesions that were significantly elevated. Three patients had mild elevation and were graded as grade 1. At the end of 6 months, all patients’ lesions showed reduction in elevation with 60% patients having flat appearance. (p value 0.0186).In group 2, at baseline 90% had elevated and one patient had mildly elevated appearance. At the end of 6 months, 30% patients still had a significantly elevated lesion and another 50% showed some reduction but persistent elevation. Only 20% patients had a flat lesion. The reduction in elevation after treatment in group 2 was not statistically significant. (p value 0.0719).
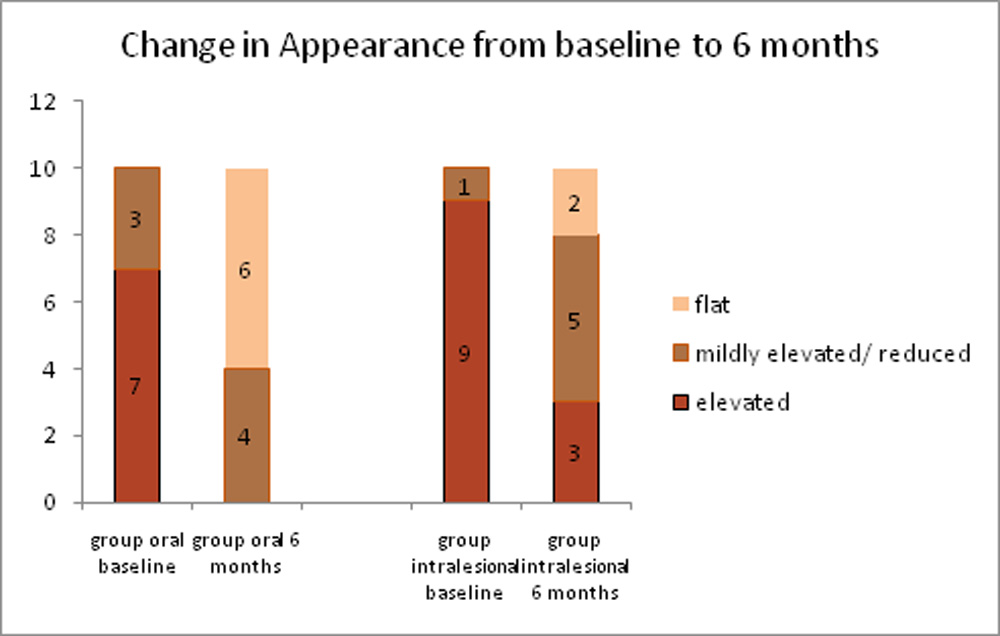
Figure 7: Graph depicting change in appearance from baseline to 6 months
e) Change in Ptosis
There was improvement in ptosis in both the groups from baseline to 6 months. At baseline, four out of six patients in group 1 had moderate ptosis and 2 had severe ptosis. At the end of 6 months three had mild ptosis of less than 2 mm and 3 had moderate ptosis. Average percentage reduction in ptosis was 66.11 ± 14.21% in group 1.
In group 2, three patients had severe ptosis at baseline, 4 had moderate and 1 had mild ptosis. At the end of six months, 2 patients continued to have severe ptosis, 1 had moderate ptosis and 5 had mild residual ptosis of less than 2 mm. The average percentage reduction in ptosis was 45.91 ± 36.52%. Two cases did not show any improvement in ptosis (percentage change at 6 months = 0%)
There was a better percentage reduction in group 1 as compared to group 2 even though the reduction was not statistically significant. All six patients with ptosis in group 1 showed improvement. As compared to this, two patients out of eight in group 2 did not show any improvement in the amount of ptosis.
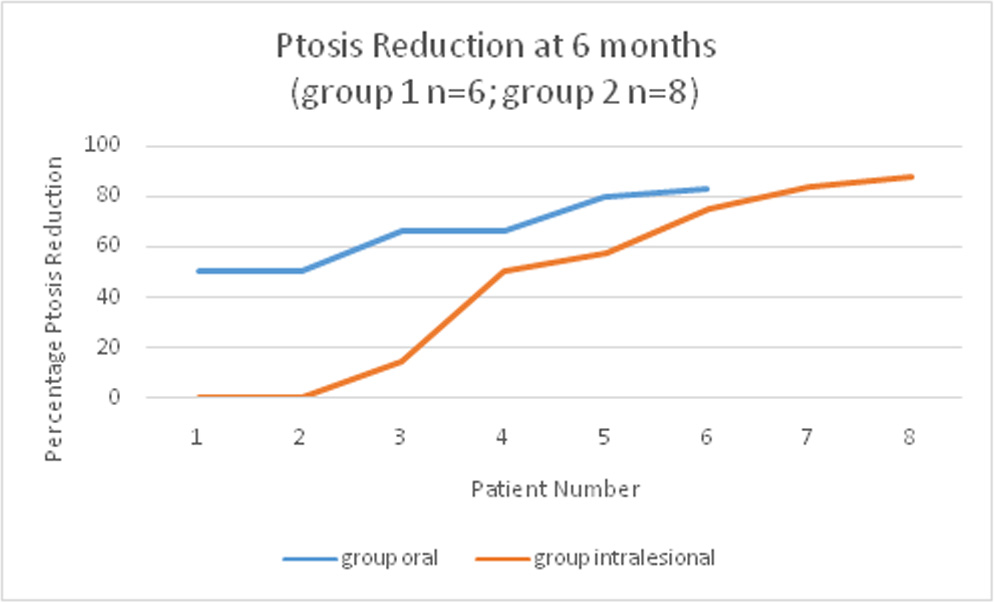
Figure 8: Graph depicting improvement in ptosis from baseline to 6 months
f) Side effects of treatment:
The parents were asked for presence of any side effects like lethargy, poor feeding, gastric reflux/ regurgitation, wheezing or irritability. Heart rate and capillary filling time were assessed at serial follow up. Specific local side effects like local ulceration or necrosis at side of haemangioma were noted. Out of group 1 (oral propranolol) 70% patients didn’t report any side effects. 20% patients noted lethargy and 10% (1 patient) noted poor feeding during the follow up period. The parents were asked to monitor the child more frequently and alter timing of evening dose of propranolol closer to bedtime. The dose was not reduced in the group and the treatment was continued as per protocol. None of the patients in group 2 (intralesional propranolol) reported any side effects, local or systemic. This difference was not statistically significant (p value 0.171).
g) Retrospective subgroup analysis within Group 2:
Within group 2 (those receiving intralesional propranolol), we retrospectively compared the baseline characteristics of the patients having excellent or very good response with an area reduction >70% at 6 months (group 2a, n=5) with the remaining patients who showed a lesser response (group 2b, n=5).
The data is summarized in the table below.
| Parameter | Group 2a (n=5) | Group 2b (n=5) | p value |
| Age (mean; median) | 10,8 | 26.2,21 | 0.1662 |
| Type (nature) of lesion | N=5 | N=5 | 0.524 |
| Area baseline (mean ±SD) | 171.07 ±106.26 mm2 | 493.536 ±308.6mm2 | 0.049 |
| Colour | N=5 | N=5 | 0.167 |
| Appearance | N=5 | N=5 | 0.9 |
Table 7: Comparison within subgroups 2a and 2b
The only statistically significantly different parameter between these two groups was area at baseline. The lesions, which showed an excellent or very good response, had a smaller size at baseline. The overall percentage area reduction in this group (group 2a) was 85.28% (range 72.65% to 92.31%). As compared to this, the overall percentage reduction in area in group 2b was 50.28% (range 31.48% to 69.1%). This difference in these two groups was statistically significant with a p value of 0.0018. Thus, small, localized lesions showed significantly better response to intralesional propranolol.
Discussion
Capillary haemangiomas are one of the most common infantile benign tumours with a reported prevalence of 0.1-0.28% in Indian studies.(1, 33)
These benign tumours are usually noted at birth, exhibit rapid postnatal growth followed by slow involution, often leading to a complete regression. Regression rate in periorbital capillary haemangiomas has been studied to be 30% by age three & 70% by age seven.(6)
Probable risk factors shown in various studies include a history of female gender, prematurity, low birth-weight, placental anomalies and multiple pregnancies.(3, 4)
In the present study, 55% patients were male and 45% were female. Their ages ranged from 3 months to 5 years (60 months). The median age was 8 months (range 3-12 months) in group 1 (oral) and 10 months (range 5-60 months) in group 2 (intralesional). Three patients had history of preterm delivery and two of them had history of twin pregnancy. Majority had history of normal vaginal delivery (15 patients). One patient had a history of low birth weight. Thus, the findings of low birth weight, history of preterm delivery and twinning were seen in some patients in the present study group. However, in the absence of a control cohort, we are unable to comment on the causative significance of these findings.
Haemangiomas can be classified as superficial, deep, or compound. The superficial lesions include red nodules without a subcutaneous component. A deep haemangioma protrudes with an overlying bluish tint or telangiectasia. Compound haemangiomas are lesions having both deep and superficial components.(34) In the present study, we classified the periorbital lesions into four main types: skin surface localized, skin surface extensive, conjunctival surface localized and deep orbital lesions. This distribution was based on clinical and MRI findings. Majority had deep extensive lesions (40%); 25% had skin surface localized lesions, 25% had conjunctival localized and another 10% had skin surface extensive lesions. The overall distribution was 12 patients with superficial lesions, 5 with deep lesions and 3 with compound lesions.
Lesions of the head and neck region often coincide with the distribution of the trigeminal nerve. Multifocal haemangiomas also exist, and infants with greater than 5 lesions should undergo workup to rule out visceral involvement.(34) We also observed the distribution of lesions and noted the site of each lesion in case a patient had more than one lesion or extensive involvement. Three patients also had associated cutaneous lesions outside the head and neck region. Where warranted, patients underwent an abdominal sonography in consultation with the paediatrician. None of the twenty patients in the present study showed any visceral lesions.
Most frequently, the lids were involved with 10 patients having an upper lid involvement and 4 patients having a lower lid involvement. Eight patients had a deep component with orbital involvement. Five patients had involvement of the eyebrow skin, 3 had lesions involving the forehead and nasal bridge and another 2 had involvement of the upper lip and cheek.
Response to treatment:
Treatment of haemangiomas has known to lead to a reduction in size as well as change in colour and appearance. Léauté-Labrèze reported that treatment with propranolol causes vasoconstriction of supplying capillaries. This reduces the blood flow within the haemangioma producing a visible change in colour and a palpable softening of the haemangioma. These effects can be observed within 1–3 days after the onset of therapy.(11)
In a review article by Tavakoli et al in 2017, the colour of periocular haemangiomas was described. Superficial lesions appearing at birth or after a few months comprised of a red papule or nodule having a flat or a rough surface and blanching with pressure. Deep lesions caused blue to purple discoloration of the skin, or may only caused anatomical disfigurement without discoloration.(35) Various case reports of treatment of periocular haemangiomas with propranolol have reported a similar reduction in colour from dark red to pale pink and resolution of telengiaectatic surface vessels. In addition to this, with reduction in size there was also flattening and change in appearance of the lesions.(36-39)
A meta-analysis comparing propranolol and other treatments (steroids, laser ablation, surgery, atenolol and timolol) for infantile haemangiomas by Liu et al reported greater efficacy and better safety profile of propranolol as compared to the other modalities.(40)
In the present study, we characterized the baseline features of the lesions in terms of surface area, colour and appearance of lesion and presence of dilated vessels. These features were subsequently evaluated at 6 months to assess the response of the lesion to the respective treatment.
Oral propranolol is highly efficacious in cutaneous infantile haemangiomas with a reported response rate of 98% (range 82-100%) as shown in a review by Marqueling et al. They reviewed 41 articles with over 1200 patients of IH receiving oral propranolol. The response rate was defined as any improvement with oral propranolol. Treatment response rates were comparable for studies evaluating IHs at specific sites, such as periorbital IHs.(23)
Qin et al performed one of the largest studies on oral propranolol in 58 infants with infantile haemangiomas. They administered oral propranolol at a dose of 1–1.5 mg/kg/day and reported a response rate of “good to excellent” in 67% of patients.(41)
Various studies have reported high efficacy of oral propranolol in periorbital haemangiomas.(37, 39, 42-48) These are summarized in table 8.
A similar efficacy with oral propranolol was also observed in the present study. All patients in group 1 (n=10) receiving oral propranolol in showed a significant response to treatment with an average percentage area reduction of 83.48% ±11.67% at the end of six months (p value 0.0019). All ten patients had more than 65% improvement in area and the response was graded as good or higher. In addition to reduction in area, there was improvement in colour and appearance of the lesions. 70% had a dark red-blue colour at baseline. At the end of 6 months, 80% had colour same as surrounding structure and 20% had a pink appearing lesion showing a significant improvement. At baseline, 70% patients in group 1 had lesions that were significantly elevated and three patients had mild elevation. At the end of 6 months, all patients’ lesions showed reduction in elevation with 60% patients having flat appearance. Dilated vessels seen at baseline in 4 out of the 10 patients became normal appearing in 3 patients at the end of 6 months.
| Author | Demographics | Dose of oral propranolol | Outcome |
| Sans et al(37)
2009 |
Number of cases: 13 (total 32, 13 periocular)
Age at enrolment: 2-41 months Duration: 3-10 months
|
2-3mg/kg/day | The efficacy of propranolol reached 100%, with the first effects appearing in the first hours with change in colour and softening of lesion
USG at 60 days showed mean regression of 40% in 11 patients |
| Li et al(42)
2010 |
Number of cases: 4
Age at enrolment: 11 weeks-2.5 years Duration: 6-12 months |
2mg/kg/day | Reduction in lesion on imaging, improvement in proptosis and astigmatism in all patients. |
| Haider et al(44)
2010 |
Number of cases: 17
Age at enrolment: 3 weeks to 12 months Duration: until resolution/ 9-11 months of age |
Initially 0.5, increased slowly to 2mg/kg/day | 10 excellent responders >50% reduction to near total resolution
6 good response moderate decrease in size 1 fair response no additional growth but no clinically significant regression |
| Cheng et al(45)
2010 |
Number of cases: 10
Age at enrolment: 3-11 months Duration of treatment: 12-42 weeks |
2mg/kg/day | Mean pre treatment area: 756.7 mm2; Mean post treatment area: 543.2 mm2
(p value 0.075) Mean pre treatment astigmatism 2.8D Mean post treatment astigmatism 1.9D |
| Claerhout et al(39)
2011 |
Number of cases: 10
Age at enrolment: 2-19 months Duration: 7.6 months mean duration |
2mg/kg/day | 5 excellent responders >50% reduction to near total resolution
3 good response moderate decrease in size 2 fair response no additional growth but no clinically significant regression |
| Harikrishna et al(46)
2011 |
Number of cases: 4
Age at enrolment: 3-19 months Duration: more than 6 months |
2mg/kg/day | Notable regression in the deeper orbital components was observed on examination and imaging in all subjects |
| Missoi et al(47) | Number of cases: 17
Age at enrolment: 2.2-5.6 months (IQR) Duration: 4.1-7.2 months (IQR)
|
2mg/kg/day | Median change in the surface area was 61% (interquartile range, 32%-64%) of the original size |
| Snir et al(43)
2011 |
Number of cases: 30
Age at enrolment: 1 to 17 months Duration: 1-15 months
|
2mg/kg/day
|
The findings revealed a mean diseased periorbital area of 12.7±21.7 cm2 before therapy and 6.0±9.5 cm2 after therapy, with a reduction of >50%
There were non-significant reductions of 31% in spherical power and 28% in mean spherical equivalent |
Continued
| Author | Demographics | Dose of oral propranolol | Outcome |
| Léauté-Labrèze et al(48)
2015 |
Number of cases: 318
(Total 456, 318 facial) Age at enrolment: 35-105 days Duration: analysis at 24 weeks |
Out of 318:
a) 40 patients placebo b) 71 patients 1mg/kg/day for 3 months c) 72 patients 1mk/kg/day for 6 months d) 64 patients: 3mg/kg/day for 3 months e) 71 patients 3mg/kg/day for 6 months |
Nearly complete resolution was defined as a minimal degree of telangiectasia, erythema, skin thickening, soft-tissue swelling, and distortion of anatomical land- marks.
Overall, 61 of 101 patients (60%) assigned to the selected propranolol regimen and 2 of 55 patients (4%) assigned to placebo had successful treatment at week 24 (P<0.001) |
| The present study | Number of cases: 10
Age at enrolment: 3-12 months Duration: 6 months |
3mg/kg/day | All 10 patients had more than 65% reduction in area percentage
In addition, there was improvement in colour and appearance, reduction in ptosis and astigmatism |
Table 8: Summary of various studies on oral propranolol in periorbital capillary haemangiomas
Only a few studies are available which discuss the role of intralesional propranolol in periocular haemangiomas. Awadein et al compared intralesional steroid with intralesional propranolol injection in periocular capillary haemangiomas. They administered a single injection of intralesional propranolol 1mg/ml concentration in a dose of 0.2ml/cm greatest linear dimension (GLD) with maximum dose of 1 ml in twelve cases. Final assessment was done at 4 months. Regression was noted in 10 out of 12 patients in terms of reduction in size, flattening of lesion and blanching of colour. At the end of the follow-up period, 42% of patients showed an excellent response with almost complete resolution of the lesion, 25% showed a good response with more than 50% reduction in the size of the lesion and 17% showed a fair response with less than 50% reduction in the size of the lesion. Only two patients (17%) were resistant to treatment. The intralesional steroid group showed regression in eight out of 10 patients with 40% patients showing an excellent response. The onset of regression was delayed in the steroid group as compared to the propranolol group.(21)
Zaher et al compared oral, topical and intralesional propranolol for problematic haemangiomas. In their study, an excellent response (complete resolution) was achieved in 9/15 patients with oral propranolol (60%), 3/15 with topical propranolol (20%) and 2/15 with intralesional propranolol (13.3%). The onset of initial response and the time taken for final response was longer in the intralesional group. As intralesional application did not offer any benefit over topical application and oral propranolol, it could not be recommended.(19) Torres-Pradilla et al studied the role of intralesional propranolol in six patients with facial capillary haemangiomas. They concluded that overall beneficial role of intralesional propranolol in infantile haemangiomas was guarded. They attributed the failure of intralesional propranolol in their study to erratic absorption, lack of a local deposit after intralesional injection, inappropriately low dose and number of injections and a higher mean age of patients in their study (7.3 months).(22)
In the present study, the response to treatment with intralesional propranolol was assessed in ten patients in group 2. At 6 months, the average percentage area reduction was 67.78% ±21.71% (p value 0.0019). 80% of the patients in group 2 had a good or higher response (>50% reduction in area percentage). Twenty per cent patients in group 2 had a fair response (30-49.9% reduction) with the least responsive patient showing only 31.48% reduction in area. 70% patients had a dark red-blue colour at baseline and 30% had red appearing lesions. At the end of 6 months, 20% patients remained to have a dark red-blue colour and 50% had a pink appearing lesion. Only 30% patients had a colour that was same as the surrounding skin. In group 2, at baseline 90% had elevated and one patient had a mildly elevated appearance. At the end of 6 months, 30% patients still had a significantly elevated lesion and another 50% showed some reduction but persistent elevation. Only 20% patients had a flat lesion. Out of the five patients with dilated vessels at baseline in group 2, three had normal appearing vessels at the end of 6 months. The results seen in the present study in patients receiving intralesional propranolol are significantly better than those reported by Torres-Pradilla et al and Zaher et al. These are summarized in Table 9.
| Author | Demographics | Dose | Outcome |
| Awadein et al(21)
2011 |
Number of cases: 12 for intralesional propranolol
10 for intralesional triamcinolone Age at enrolment: Propranolol: 5.9 ±2.7 months Triamcinolone: 6.1±2.9 months Duration: 4 months follow up
|
Propranolol 1mg/ml
Triamcinolone 40mg/ml
Single injection 0.2ml/cm GLD, maximum of 1 ml |
Propranolol group:
Pre treatment size: 8.1 ±3.4, range 3.6–14.3 cm2; Post treatment size: 3.6 ±2.6, range 0– 7.1 cm2; 10/12 showed regression Steroid group: Pre treatment size: 7.9 ±3.6, range 3.5–14.1 cm2; Post treatment size: 3.7 ±2.5, range 0–7.5 cm2; 8/10 showed regression |
| Torres-Pradilla et al(22)
2013 |
Number of cases: 6
Age at enrolment: 7.3±4.3 months Duration: 7.3 months |
1mg/ml, 0.2ml/cm of GLD
Injection repeated if response seen at 1 month; 1 patient: 1 injection; 4 patients: 2 injections; 1 patient: 3 injections |
No significant response in terms of regression in size, colour or appearance |
| Zaher et al(19)
2013 |
Number of cases: 45 (15 in each group)
Age at enrolment: Group A: 3-18 months Group B: 1-18 months Group C: 3-18 months Duration: Group A: 3-9 months Group B: 5-10 months Group C: 5-12 months Analysis at 6 months after completing treatment |
Group A: oral propranolol 2mg/kg/day
Group B: topical propranolol 1% ointment Group C: intralesional propranolol 1mg/ml 0.2ml/GLD weekly injections |
Improvement noted in 86.7% patients in group A, 66.7% patients in group B and 53.3% patients in group C
Recommendations: First line of treatment of IH is oral propranolol. Topical propranolol could be considered in patients at risk of potential side effects from oral administration. No benefit with intralesional propranolol hence not recommended. |
Continued
| Author | Demographics | Dose | Outcome |
| The present study | Number of cases: 10
Age at enrolment: 5-60 months Duration of follow up: 6 months |
1mg/ml propranolol hydrochloride
0.2ml/cm of longest linear dimension (maximum of 1 ml) 3 injections 4-6 weeks apart |
An average area reduction of 67.78±21.71% (p value 0.0019)
8 out of 10 had >50% reduction in size In addition, there was improvement in colour and appearance, reduction in ptosis and astigmatism |
Table 9: Summary of various studies on intralesional propranolol in capillary haemangiomas
A further analysis in comparing the reduction in percentage area between groups 1 and 2 in the present study revealed no statistically significant difference in response between oral and intralesional propranolol at 6 months (p value 0.056). Thus, intralesional propranolol seemed comparable to oral propranolol therapy at the six months analysis in the present study.
Associated Complications
Although most infantile haemangiomas are usually not problematic, up to 12% can cause significant morbidity, including disfigurement, difficulty in feeding, ulceration, vision loss, airway compromise, congestive heart failure, and death.(9, 49-53)
A study focusing on periocular haemangiomas done by Haik et al reported that complications could occur in up to 80% of untreated or, alternatively, treatment-resistant periocular capillary haemangioma cases.(54) Even though most lesions regress spontaneously, treatment is however indicated if there is an obstruction of the visual axis, induced astigmatism, strabismus, exposure keratitis, rapid growth, optic nerve compression, severe ptosis or poor cosmesis.(51, 55-59) Amblyopia is the primary concern because of stimulus deprivation, induced anisometropia (mainly oblique astigmatism), secondary strabismus, or globe displacement.(60)
Anisometropia induced amblyopia, the most significant sequelae in terms of frequency, affects up to 60% of these patients.(8,54) This form of amblyopia is usually the result of mass-induced indentation on the infant’s flexible sclera and cornea, causing distortion and astigmatism.(9,51) Deprivation amblyopia resulting from obstruction of the visual axis by the lesion is an additional potential cause for amblyopia. A study on refractive and structural changes of periorbital haemangiomas by Snir et al reported the association of anisometropic astigmatism, refractive amblyopia and ptosis – induced amblyopia in periocular haemangiomas. They treated 30 patients with oral propranolol 2mg/kg/day for a mean duration of 7.3±3.5 months. They reported a reduction of 40.5% in mean cylindrical power in the involved eye after treatment compared with baseline (p value 0.02). There was a non-significant reduction of 31% in spherical power (p value 0.13), and a non-significant decrease of 28% in mean spherical equivalent (p value 0.1). In their study, in addition to oral propranolol, patients with significant refractive anisometropia (mainly astigmatism) or partial occlusion of the visual axis were prescribed glasses; those older than 4 months (3 patients) were also given anti amblyopia treatment (patching).(43)
In a study by Fabian et al, the role of oral propranolol 2mg/kg/day for the reduction of astigmatism was assessed in three cases of periocular haemangiomas. The mean astigmatic error decreased from 2.83 Dioptres (range 2.5-3.0D) before propranolol treatment to 1.33 dioptres (range 1.0-2.0D) after 1 month of treatment. A rapid therapeutic effect was noticed in all cases, including a change in lesion size and colour.(60) Herlihy et al studied the effect of oral propranolol treatment on visual acuity and astigmatism in 17 patients of periocular haemangiomas. Mean astigmatism in affected eyes was 1.90 D pre-treatment and 1.00 D post-treatment (p value 0.0033), and 0.69 D and 0.40 D, respectively, in unaffected eyes (p value 0.19). Patients showed a monophasic reduction in astigmatism over 12 months. None of the patients had visual acuity in the affected eye more than 1 standard deviation below the age- matched norm. No patient experienced significant side effects when treated with oral propranolol.(61)
In the present study, keratometry was recorded in all patients and where possible, visual acuity was also documented. Analysis of corneal astigmatism was done at baseline and at 6 months. The mean pre treatment corneal astigmatism in group 1 was 2.86D at 179.8 degrees. There was a 60.53% reduction in amount of astigmatism and mean post treatment astigmatism was 1.13D at 179.81 degrees; p value 0.0045. The mean pre treatment astigmatism in group 2 was 1.62D at 90.16 degrees. There was a 53.54% reduction in amount of astigmatism and mean post treatment astigmatism was 0.75D at 179.87 degrees; p value 0.0001. On comparing the amount of reduction in dioptres of astigmatism between the two groups, there was no significant difference; p value 0.49. Thus both groups showed a significant reduction in astigmatism with treatment and the reduction was comparable between the two treatment groups.
Regression in size of lesion results in improvement of ptosis especially for upper eyelid lesions. Severe ptosis is one of the indications for treating periocular haemangiomas. Harikrishna et al have studied the effect of oral propranolol and reduction of ptosis from baseline in four patients.(46) In the present study, fourteen out of 20 patients had some degree of ptosis at baseline. At baseline, four out of six patients in group 1 had moderate ptosis (2-4mm) and 2 had severe ptosis (>4mm). At the end of 6 months three had mild ptosis of less than 2 mm and 3 had moderate ptosis. Average percentage reduction in ptosis was 66.11 ±14.21% in group 1. In group 2, three patients had severe ptosis at baseline, 4 had moderate and 1 had mild ptosis. At the end of six months, 2 patients continued to have severe ptosis, 1 had moderate ptosis and 5 had mild residual ptosis of less than 2 mm. The average percentage reduction in ptosis was 45.91 ±36.52%. Two cases in group 2 did not show any improvement in ptosis. The reduction in ptosis was as a result in reduction in overall size of the lesion. Other complications assessed in the present study were squint (none), presence of ulceration or necrosis (none) and systemic evaluation and presence of any cardiorespiratory or abdominal complications (none).
Side Effects of Therapy
Side effects of systemic propranolol have been evaluated in many studies. Propranolol is thought to cause hypoglycaemia by inhibiting glycogenolysis, gluconeogenesis and lipolysis. Children seem to be more susceptible to hypoglycaemia as they have lower glycogen stores leading to a reduced fasting ability and a higher glucose utilisation rate. In addition, beta blockers mask the early sympathetic signs of hypoglycaemia like tachycardia, sweating and palpitations. In a large systematic review of IHs treated with propranolol, Marqueling et al. reported the most common adverse events as changes in sleep and acrocyanosis seen in 11.4% and 5.1% patients and rare incidence of serious adverse events such as symptomatic hypotension in 0.4%, hypoglycaemia in 0.3%, and symptomatic bradycardia in 0.08% patients, respectively.(23) In a review on oral propranolol by Cornish et al in 2011, adverse effects with propranolol for infantile periocular capillary haemangiomas were documented in 26 of the 100 cases. However, most of these were not serious enough to warrant stopping or amending the dose of propranolol; propranolol was only discontinued in five cases.(62) A meta-analysis comparing propranolol and other treatments (steroids, laser ablation, surgery, atenolol and timolol) for infantile haemangiomas by Liu et al reported greater efficacy and better safety profile of propranolol as compared to the other modalities. Their review included 61 studies in which 30 studies with a total of 1893 patients were based on oral propranolol. Out of these 1893 patients, side effects were reported in 286 (15.11%) patients only. These included insomnia, hypotension, respiratory disorder, diarrhoea, cold extremities, fatigue, hypoglycaemia, constipation, and other gastrointestinal disturbances.(40) Various studies have reported side effects with use of oral propranolol for treatment of capillary haemangiomas.(37,39,42-48) These are summarized in table 10.
In the present study, in group 1 70% patients didn’t report any side effects. 20% patients noted lethargy and 10% (1 patient) noted poor feeding during the follow up period. The parents were asked to monitor the child more frequently and alter timing of evening dose of propranolol closer to bedtime. The dose was not reduced in the group and the treatment was continued as per protocol.
| Author | Number of patients | Dose of oral propranolol | Number of side effects reported | Side effects |
| Sans et al(37) 2009 | 13 | 2-3mg/kg/day | 1 | Wheezing |
| Li et al(42) 2010 | 4 | 2mg/kg/day | none | |
| Haider et al(44)
2010 |
17 | 2mg/kg/day | 6 | Gastrointestinal upset, intermittent fatigue, spitting, shaking episodes |
| Cheng et al(45) 2010 | 10 | 2mg/kg/day | none | |
| Claerhout et al(39) 2011 | 10 | 2mg/kg/day | 1 | Wheezing |
| Harikrishna et al(46) 2011 | 4 | 2mg/kg/day | none | |
| Missoi et al(47) 2011 | 17 | 2mg/kg/day | 1 | Bradycardia |
| Snir et al(43) 2011 | 30 | 2mg/kg/day
|
11 (treatment discontinued in 1, dose reduced in 3) | Respiratory tract infection, breathlessness, loss of appetite, diarrhoea, vomiting, sleep problems, restlessness, fever |
| Léauté-Labrèze et al(48) 2015 | 456 (318 facial haemangiomas) | a) 55 patients placebo
b) 98 patients 1mg/kg/day for 3 months c) 102 patients 1mk/kg/day for 6 months d) 100 patients: 3mg/kg/day for 3 months e) 101 patients 3mg/kg/day for 6 months |
26 patients had serious adverse events
a) 3 b) 5 c) 3 d) 9 e) 6
|
Hypotension, bradycardia, bronchospasm, hypoglycaemia
Others: diarrhoea, sleep disturbance, agitation, vomiting, cold extremities, constipation, decreased appetite |
| The present study | 10 | 3mg/kg/day | 3 | Lethargy, poor feeding |
Table 10: Summary of studies on side effects of oral propranolol in treating capillary haemangiomas
A research workshop in 2005 on paediatric dermatology discussed an equivalent efficacy with intralesional corticosteroid injections as compared to systemic corticosteroids. This study cautioned regarding the rare risk of inadvertent intravascular/ophthalmic artery embolization with permanent visual loss during administration.(30) The most serious and feared complication is central retinal artery occlusion (CRAO).(29) Bang et al recommended that while administering intralesional corticosteroid, retinal vessels should be examined during and after injection to monitor for central retinal artery occlusion. It has been speculated that increased force from the injection or digital pressure after the procedure may cause retrograde flow of the drug particles into the central retinal artery.(18) Though this has been documented with steroid injection, it may be a potential risk with intralesional propranolol also. Awadein et al compared intralesional propranolol injection and intralesional triamcinolone in periocular capillary haemangiomas and they did not report any adverse effects during or after intralesional propranolol injection.(21)
In the present study, none of the ten patients in group 2 (intralesional propranolol) reported any side effects, local or systemic. All the injections were administered under general anaesthesia with cardiorespiratory monitoring. As compared to the study by Zaher et al, pain was not a reported side effect both during the injection procedure and subsequently in the observation period.(19) None of the patients in the present study reported any side effects related to the general anaesthesia. However, it is pertinent to highlight the potential side effects and the need for general anaesthesia for the procedure, given the age group of these patients. Older children may be cooperative for the procedure under topical anaesthesia. Alternatively, the procedure may be undertaken under sedation with peri-operative analgesia. This may add to the logistics of the procedure. In addition, pre and post anaesthesia care is required with monitoring of systemic parameters including heart rate and respiratory rate. These are summarised in table 11.
| Author | Number of patients | Dose of intralesional propranolol | Number of side effects reported | Side effects |
| Awadein et al(21)
2011 |
12 | 1mg/ml
Single injection 0.2ml/cm GLD, maximum of 1ml Dilated fundus exam done during injection, administered under general anaesthesia |
None | |
| Zaher et al(19)
2013 |
15 | 1mg/ml 0.2ml/GLD weekly injections administered 60 min after topical anaesthesia with lidocaine | 3 | Severe pain during injection |
| Torres-Pradilla et al(22)
2013 |
6 | 1mg/ml, 0.2ml/cm of GLD
Injection repeated if response seen at 1 month |
6
|
Pain (all patients), erythema |
| The present study | 10 | 1mg/ml propranolol hydrochloride
0.2ml/cm of longest linear dimension (maximum of 1 ml) 3 injections 4-6 weeks apart, administered under general anaesthesia |
None |
|
Table 11: Summary of studies on side effects of intralesional propranolol in treating capillary haemangiomas
To summarise, in contrast to two studies published by Zaher et al and Torres-Pradilla et al, in the present study we found a statistically comparable response at 6 months with intralesional propranolol as compared to oral propranolol.(19,22) Within the group receiving intralesional propranolol, we retrospectively compared the baseline characteristics of the patients having excellent or very good response with an area reduction >70% at 6 months (group 2a, n=5) with the remaining patients who showed a lesser response (group 2b, n=5). The lesions, which showed an excellent or very good response, had a smaller size at baseline. The overall percentage area reduction in this group (group 2a) was 85.28% (range 72.65% to 92.31%). As compared to this, the overall percentage reduction in area in group 2b was 50.28% (range 31.48% to 69.1%). This difference in these two groups was statistically significant with a p value of 0.0018. Thus, small, localized lesions showed significantly better response to intralesional propranolol.
Studies to analyse local concentration of drug after intralesional administration and detailed histopathological analysis to assess effect of injection procedure on the lesion can help better delineate the mode of action of intralesional propranolol and highlight its differences as compared to oral propranolol. In addition, development of a depot formulation of propranolol for intralesional administration may help in improving its efficacy.
The response seen in the present study in both groups was attributed to the treatment intervention. It could have however occurred as a part of the natural course of the regression of the haemangioma. The median age in the present study was 8 months (range 3-12 months) in group 1 and 10 months (range 5-60 months). Thus majority of the lesions were in the proliferating phase. However, in the absence of a control observation-only group, we can’t comment conclusively on this finding.
Summary
This was a prospective randomized control trial with 20 patients, 10 received oral propranolol (group 1) and 10 received intralesional propranolol (group 2). The baseline characteristics were comparable in both groups (except gender). Serial follow up was maintained and the final evaluation was done at 6 months.
Both groups responded well to treatment in terms of percentage area reduction. The average percentage area reduction in group 1 (oral propranolol) was 83.48% ±11.67% at the end of six months (p value 0.0019). The average percentage area reduction in group 2 (intralesional propranolol) was 67.78% ±± 21.71% (p value 0.0019). There was no significant difference between the comparative reduction in percentage area in group 1 and 2 (p value: 0.056)
There was a significant change in colour of the lesions in each group (p value group 1: 0.04 and group 2: 0.02). There was a statistically significant greater improvement in colour in group 1 as compared to group 2 (p value: 0.021). There was no significant difference in the comparative reduction in appearance (from elevated to flat; p value 0.085) and reduction in ptosis (severe to mild; p value: 0.23) between the two groups. There was a 60.53% reduction in dioptric amount of astigmatism in group 1 and a 53.54% reduction in dioptric amount of astigmatism in group 2. (p values: Group 1: 0.0045 and Group 2: 0.0001). Thus both groups showed a significant reduction in astigmatism with treatment and the reduction was comparable between the two treatment groups (p value 0.49) Within group 2, small, localized lesions showed significantly better response to intralesional propranolol as compared to larger lesions.
No patient reported any side effects in the intralesional group in the present study compared to 3 patients having lethargy/ poor feeding in the oral group (p value 0.171, statistically non-significant).
Thus, intralesional propranolol may be considered a viable alternative to oral propranolol for periocular capillary haemangiomas. It may be considered as a first line therapy in small surface localized capillary haemangiomas. It may also be considered an alternate treatment in patients already on oral propranolol showing significant systemic side effects. A randomised control trial with a larger sample size, an observation only control group and a longer follow up should be done to conclusively determine the beneficial role of intralesional propranolol.
References
- Nanda A, Kaur S, Bhakoo ON, Dhall K. Survey of cutaneous lesions in Indian newborns. Pediatr Dermatol. 1989;6(1):39-42.
- Cruz OA, Siegfried EC. Propranolol treatment for periocular capillary hemangiomas. J AAPOS. 2010;14(3):199-200.
- Haggstrom AN, Drolet BA, Baselga E, Chamlin SL, Garzon MC, Horii KA, et al. Prospective study of infantile hemangiomas: demographic, prenatal, and perinatal characteristics. J Pediatr. 2007;150(3):291-4.
- Munden A, Butschek R, Tom WL, Marshall JS, Poeltler DM, Krohne SE, et al. Prospective study of infantile haemangiomas: incidence, clinical characteristics and association with placental anomalies. Br J Dermatol. 2014;170(4):907-13.
- Mulliken JB, Glowacki J. Hemangiomas and vascular malformations in infants and children: a classification based on endothelial characteristics. Plast Reconstr Surg. 1982;69(3):412-22.
- Kanski JJ, Bowling B, Nischal KK, Pearson A. Clinical ophthalmology : a systematic approach. 7th ed. Edinburgh ; New York: Elsevier/Saunders; 2014. ix, 909 p. p.
- Tambe K, Munshi V, Dewsbery C, Ainsworth JR, Willshaw H, Parulekar MV. Relationship of infantile periocular hemangioma depth to growth and regression pattern. J AAPOS. 2009;13(6):567-70.
- Stigmar G, Crawford JS, Ward CM, Thomson HG. Ophthalmic sequelae of infantile hemangiomas of the eyelids and orbit. Am J Ophthalmol. 1978;85(6):806-13.
- Cuttone JM, Durso F, Miller M, Evans LS. The relationship between soft tissue anomalies around the orbit and globe and astigmatic refractive errors: a preliminary report. J Pediatr Ophthalmol Strabismus. 1980;17(1):29-36.
- Leaute-Labreze C, Harper JI, Hoeger PH. Infantile haemangioma. Lancet. 2017.
- Leaute-Labreze C, Dumas de la Roque E, Hubiche T, Boralevi F, Thambo JB, Taieb A. Propranolol for severe hemangiomas of infancy. N Engl J Med. 2008;358(24):2649-51.
- Storch CH, Hoeger PH. Propranolol for infantile haemangiomas: insights into the molecular mechanisms of action. Br J Dermatol. 2010;163(2):269-74.
- Chim H, Armijo BS, Miller E, Gliniak C, Serret MA, Gosain AK. Propranolol induces regression of hemangioma cells through HIF-1alpha-mediated inhibition of VEGF-A. Ann Surg. 2012;256(1):146-56.
- Ji Y, Li K, Xiao X, Zheng S, Xu T, Chen S. Effects of propranolol on the proliferation and apoptosis of hemangioma-derived endothelial cells. J Pediatr Surg. 2012;47(12):2216-23.
- Sommers Smith SK, Smith DM. Beta blockade induces apoptosis in cultured capillary endothelial cells. In Vitro Cell Dev Biol Anim. 2002;38(5):298-304.
- Ames JA, Sykes JM. Current trends in medical management of infantile hemangioma. Curr Opin Otolaryngol Head Neck Surg. 2015;23(4):286-91.
- Pierre Fabre Pharmaceuticals I. HIGHLIGHTS OF PRESCRIBING INFORMATION for Hemangeol. [Internet]. 2014. [cited February 13th 2017]. Available from: https://www.accessdata.fda.gov/drugsatfda_docs/label/2014/205410s000lbl.pdf
- Bang GM, Setabutr P. Periocular capillary hemangiomas: indications and options for treatment. Middle East Afr J Ophthalmol. 2010;17(2):121-8.
- Zaher H, Rasheed H, Esmat S, Hegazy RA, Gawdat HI, Hegazy RA, et al. Propranolol and infantile hemangiomas: different routes of administration, a randomized clinical trial. Eur J Dermatol. 2013;23(5):646-52.
- Hao J, Yang MB, Liu H, Li SK. Distribution of propranolol in periocular tissues: a comparison of topical and systemic administration. J Ocul Pharmacol Ther. 2011;27(5):453-9.
- Awadein A, Fakhry MA. Evaluation of intralesional propranolol for periocular capillary hemangioma. Clin Ophthalmol. 2011;5:1135-40.
- Torres-Pradilla M, Baselga E. Failure of intralesional propranolol in infantile hemangiomas. Pediatr Dermatol. 2014;31(2):156-8.
- Marqueling AL, Oza V, Frieden IJ, Puttgen KB. Propranolol and infantile hemangiomas four years later: a systematic review. Pediatr Dermatol. 2013;30(2):182-91.
- de Graaf M, Breur JM, Raphael MF, Vos M, Breugem CC, Pasmans SG. Adverse effects of propranolol when used in the treatment of hemangiomas: a case series of 28 infants. J Am Acad Dermatol. 2011;65(2):320-7.
- Holland KE, Frieden IJ, Frommelt PC, Mancini AJ, Wyatt D, Drolet BA. Hypoglycemia in children taking propranolol for the treatment of infantile hemangioma. Arch Dermatol. 2010;146(7):775-8.
- Kallen RJ, Mohler JH, Lin HL. Hypoglycemia: a complication of treatment of hypertension with propranolol. Clin Pediatr (Phila). 1980;19(8):567-8.
- Xu DP, Cao RY, Xue L, Sun NN, Tong S, Wang XK. Treatment of severe infantile hemangiomas with propranolol: an evaluation of the efficacy and effects of cardiovascular parameters in 25 consecutive patients. J Oral Maxillofac Surg. 2015;73(3):430-6.
- Starkey E, Shahidullah H. Propranolol for infantile haemangiomas: a review. Arch Dis Child. 2011;96(9):890-3.
- Shorr N, Seiff SR. Central retinal artery occlusion associated with periocular corticosteroid injection for juvenile hemangioma. Ophthalmic Surg. 1986;17(4):229-31.
- Frieden IJ, Haggstrom AN, Drolet BA, Mancini AJ, Friedlander SF, Boon L, et al. Infantile hemangiomas: current knowledge, future directions. Proceedings of a research workshop on infantile hemangiomas, April 7-9, 2005, Bethesda, Maryland, USA. Pediatr Dermatol. 2005;22(5):383-406.
- Retzlaff J, Paden PY, Ferrell L. Vector analysis of astigmatism. Adding and subtracting spherocylinders. J Cataract Refract Surg. 1993;19(3):393-8.
- Holladay JT, Moran JR, Kezirian GM. Analysis of aggregate surgically induced refractive change, prediction error, and intraocular astigmatism. J Cataract Refract Surg. 2001;27(1):61-79.
- Mendiratta V, Jabeen M. Infantile hemangioma: an update. Indian J Dermatol Venereol Leprol. 2010;76(5):469-75.
- Richter GT, Friedman AB. Hemangiomas and vascular malformations: current theory and management. Int J Pediatr. 2012;2012:645678.
- Tavakoli M, Yadegari S, Mosallaei M, Aletaha M, Salour H, Lee W. Infantile periocular hemangioma. Journal of Ophthalmic and Vision Research. 2017;12(2):205-11.
- Vohra V, Gupta P, Malik PK, Pathak A. Propranolol therapy in a case of capillary hemangioma. Oman J Ophthalmol. 2015;8(3):191-3.
- Sans V, de la Roque ED, Berge J, Grenier N, Boralevi F, Mazereeuw-Hautier J, et al. Propranolol for severe infantile hemangiomas: follow-up report. Pediatrics. 2009;124(3):e423-31.
- Qayyum S. Role of Propranolol in the Management of Periocular Infantile Hemangioma. Pak J Ophthalmol. 2016;32(2):7.
- Claerhout I, Buijsrogge M, Delbeke P, Walraedt S, De Schepper S, De Moerloose B, et al. The use of propranolol in the treatment of periocular infantile haemangiomas: a review. Br J Ophthalmol. 2011;95(9):1199-202.
- Liu X, Qu X, Zheng J, Zhang L. Effectiveness and Safety of Oral Propranolol versus Other Treatments for Infantile Hemangiomas: A Meta-Analysis. PLoS One. 2015;10(9):e0138100.
- Qin ZP, Liu XJ, Li KL, Zhou Q, Yang XJ, Zheng JW. [Treatment of infantile hemangiomas with low-dose propranolol: evaluation of short-term efficacy and safety]. Zhonghua Yi Xue Za Zhi. 2009;89(44):3130-4.
- Li YC, McCahon E, Rowe NA, Martin PA, Wilcsek GA, Martin FJ. Successful treatment of infantile haemangiomas of the orbit with propranolol. Clin Exp Ophthalmol. 2010;38(6):554-9.
- Snir M, Reich U, Siegel R, Zvulunov A, Friling R, Goldenberg-Cohen N, et al. Refractive and structural changes in infantile periocular capillary haemangioma treated with propranolol. Eye (Lond). 2011;25(12):1627-34.
- Haider KM, Plager DA, Neely DE, Eikenberry J, Haggstrom A. Outpatient treatment of periocular infantile hemangiomas with oral propranolol. J AAPOS. 2010;14(3):251-6.
- Cheng JF, Gole GA, Sullivan TJ. Propranolol in the management of periorbital infantile haemangioma. Clin Exp Ophthalmol. 2010;38(6):547-53.
- Harikrishna B, Ganesh A, Al-Zuahibi S, Al-Jabri S, Al-Waily A, Al-Riyami A, et al. Oral propranolol for the treatment of periorbital infantile hemangioma: a preliminary report from oman. Middle East Afr J Ophthalmol. 2011;18(4):298-303.
- Missoi TG, Lueder GT, Gilbertson K, Bayliss SJ. Oral propranolol for treatment of periocular infantile hemangiomas. Arch Ophthalmol. 2011;129(7):899-903.
- Léauté-Labrèze C, Hoeger P, Mazereeuw-Hautier J, Guibaud L, Baselga E, Posiunas G, et al. A Randomized, Controlled Trial of Oral Propranolol in Infantile Hemangioma. N Engl J Med. 2015;372(8):735-46.
- Kopf AW, Bart RS. Tumor Conference #48. Massive congenital hemangioma resulting in death. J Dermatol Surg Oncol. 1983;9(7):509-12.
- Broeks IJ, Hermans DJ, Dassel AC, van der Vleuten CJ, van Beynum IM. Propranolol treatment in life-threatening airway hemangiomas: a case series and review of literature. Int J Pediatr Otorhinolaryngol. 2013;77(11):1791-800.
- Robb RM. Refractive errors associated with hemangiomas of the eyelids and orbit in infancy. Am J Ophthalmol. 1977;83(1):52-8.
- Parikh SR, Darrow DH, Grimmer JF, Manning SC, Richter GT, Perkins JA. Propranolol use for infantile hemangiomas: American Society of Pediatric Otolaryngology Vascular Anomalies Task Force practice patterns. JAMA Otolaryngol Head Neck Surg. 2013;139(2):153-6.
- Luu M, Frieden IJ. Haemangioma: clinical course, complications and management. Br J Dermatol. 2013;169(1):20-30.
- Haik BG, Jakobiec FA, Ellsworth RM, Jones IS. Capillary hemangioma of the lids and orbit: an analysis of the clinical features and therapeutic results in 101 cases. Ophthalmology. 1979;86(5):760-92.
- Schwartz SR, Blei F, Ceisler E, Steele M, Furlan L, Kodsi S. Risk factors for amblyopia in children with capillary hemangiomas of the eyelids and orbit. J AAPOS. 2006;10(3):262-8.
- Weiss AH, Kelly JP. Reappraisal of astigmatism induced by periocular capillary hemangioma and treatment with intralesional corticosteroid injection. Ophthalmology. 2008;115(2):390-7 e1.
- Mitchell DE, Freeman RD, Millodot M, Haegerstrom G. Meridional amblyopia: evidence for modification of the human visual system by early visual experience. Vision Res. 1973;13(3):535-58.
- Levi M, Schwartz S, Blei F, Ceisler E, Steele M, Furlan L, et al. Surgical treatment of capillary hemangiomas causing amblyopia. J AAPOS. 2007;11(3):230-4.
- Wasserman BN, Medow NB, Homa-Palladino M, Hoehn ME. Treatment of periocular capillary hemangiomas. J AAPOS. 2004;8(2):175-81.
- Fabian ID, Ben-Zion I, Samuel C, Spierer A. Reduction in astigmatism using propranolol as first-line therapy for periocular capillary hemangioma. Am J Ophthalmol. 2011;151(1):53-8.
- Herlihy EP, Kelly JP, Sidbury R, Perkins JA, Weiss AH. Visual acuity and astigmatism in periocular infantile hemangiomas treated with oral beta-blocker versus intralesional corticosteroid injection. J AAPOS. 2016;20(1):30-3.
- Spiteri Cornish K, Reddy AR. The use of propranolol in the management of periocular capillary haemangioma–a systematic review. Eye (Lond). 2011;25(10):1277-83.



Leave a Comment