Dr. Radha K. Annamalai, A14830, Dr. Muthayya Muthukumar
![]()
Purpose:
The primary objective is to identify the type and clinical nature of uveitis in Hansen’s disease after treatment is completed and is proven to be negative on histopathological examination of skin biopsy.
Methods:
A prospective cohort study conducted over 3 years on of 120 patients who had completed treatment for Hansen’s disease. Complete ophthalmic evaluation, PCR and RT- PCR was done on aqueous humor samples. Chi square analysis was performed. This is a prospective study performed in a tertiary referral hospital in India. It was performed after institutional ethics committee approval and an informed consent was obtained from all patients. Inclusion criteria comprised of all patients with Hansen’s disease who had completed treatment for either paucibacillary, multibacillary or indeterminate type of leprosy. Patients who had not completed treatment and those with other forms of systemic infections such as tuberculosis, toxocariasis, candidaretinochoroiditis were excluded from the study. All cases were referred from a leprosy rehabilitation centre in Chennai, Tamilnadu and the study was conducted in concurrence with the department of dermatology. Other criteria for enrolment in the study were, adequate follow-up for atleast one year from the onset of uveitis and completeness of the medical and ocular records. The ocular examination included a visual acuity examination, slit-lamp examination, applanation tonometry, corneal sensation and Schirmer’s tests. The treatment for multibacillary leprosy was a standard regimen for 12 months consisting of rifampicin 600mg once a month, dapsone 100mg daily and clofazamine 300mg once a month. Those with paucibacillary leprosy had completed a regimen of 6 months duration which included rifampicin 600mg once a month and dapsone 100mg daily. Management of uveitis was done using topical cycloplegics and corticosteroid eye drops to manage inflammation. A split skin microscopy was performed in each patient to confirm the activity of the disease, the extent of systemic disease and the response to treatment. The smear estimated the number of acid-fast bacilli that were detected and this was reported as the bacterial Index. Skin smears were taken from earlobes, elbows and knees as well as from lesions in the patient. AC paracentesis was performed under aseptic precautions using povidone iodine and a 26 gauge needle mounted on a tuberculin syringe. 0.1ml of aqueous humour sample was obtained. ZiehlNeelsonCarbolFuschin stain was used for the diagnosis after the slide was prepared. In those with recalcitrant uveitis, a polymerase chain reaction (PCR) was performed on the aqueous humor sample. The sequences that were targeted using PCR included genes encoding the DNA of 36-kDa antigen, 18-kDa antigen, 65-kDa antigen and the repetitive sequences among other M. leprae genes. RT- PCR was performed based on response to uveitis and the aqueous humor sample of 3 patients was analysed for pathogen. Apart from genetic sequencing, complete blood counts, purified protein tests and chest x rays were done for all patients. Follow up was done for 3 years and during each visit a complete ophthalmic evaluation was performed.
Results: Incidence of uveitis was 39% with blindness in 9% which was reversible in 6%.This study was performed on 120 eyes of 90 patients with uveitis over a three year period. Ages ranged from 25 to 60 years. Acute anterior uveitis was seen in 28 % and chronic in 72%. 43% were lepromatous, 12% indeterminate and 45% tuberculoid leprosy. RT- PCR detected live bacilli in tuberculoid leprosy.
38% showed acute anterior uveitis and 62% showed chronic anterior uveitis. It was granulomatous in 83% with the predominant clinical feature being iris sphincter atrophy and dilated pupil with large mutton keratic precipitatesand anterior chamber cells and flare. 17 % of patients presented with a non- granulomatous iridocyclitis although that is not a usual presentation in uveitis. Cataract was noted in 33% of patients. On morphological evaluation, posterior subcapsular cataract was the most common feature and both senile and complicated cataract were seen. Several of our patients had dilated pupil due to iris sphincter atrophy. Vitritis was present in 3% of patients and was seen as grade 2 cells with vitreous haze. Fundus examination was normal in all patients. 12% of patients had scleritis of the nodular type. Those with scleritis showed resolution with topical steroids with no recurrence and a scraping was not performed on them. Treatment for iridocyclitis was 1% atropine eye drops,1% prednisolone acetate eye drops and oral steroids in the dose of 1mg per kg bodyweight In those with no resolution to this treatment for more than 3 weeks, an anterior chamber paracentesis was performed. RT- PCR performed on the aqueous humour of 12 patients with recalcitrant uveitis showed detection of DNA of M. leprae in 3 of them. A smear in these patients showed the presence of live and dead bacilli. Microscopy showed the typical morphology of fully formed live bacillus and beaded dead bacilli both existing in the same sample. After completion of treatment, the onset of uveitis in the paucibacillary type occurred within 1 year in 9% of patients, 2 years in 33% and within 3 years in 11%. Those with multibacillary leprosy had recurrence of uveitis after 3 years of completion of treatment. No patients had uveitis before that time period.
Conclusion: Acute anterior uveitis in leprosy is more common in the tuberculoid type despite completion of treatment. RT- PCR on aqueous is useful in chronic uveitis and requires ocular follow up even if histopathology is negative for the bacillus.Evaluation and analysis of aqueous humour by PCR has shown that DNA assays can be very sensitive in identifying bacilli and their DNA. Patients who have completed treatment are more likely to have persisting M. leprae bacilli in the aqueous humour in the paucibacillary type. Also the recurrence of uveitis is earlier in these patients than the multibacillary type. We found in our study that the commonest cause of defective vision is chronic uveitis with complicated cataract which is treatable if detected early. It may be required to start anti leprosy treatment again when live bacilli are seen in the aqueous humour even if the systemic status has settled. PCR can certainly ascertain the diagnosis by detecting even few bacilli from a small sample. This may actually be helpful in breaking the chain of leprosy transmission. PCR of aqueous humour can be adopted as a routine in all patients who present with anterior uveitis after completion of leprosy treatment. The prevalence of uveitis and blindnessin leprosy can vary between different populations. A programme for screening of leprosy should continue throughout the course of treatment and at regular intervals even after completion of treatment for both multibacillary and paucibacillary forms.
Inferences:
- RT- PCR identifies live bacilli in recalcitrant uveitis
- It is more common in the tuberculoid variety
- Longer duration of treatment may be required though it is paucibacillary type
- Recurrence of uveitis is earlier and more frequent in this type
- Middle aged and elderly develop more severe uveitis
REFERENCES
[1]. Grzybowski A, Nita M, Virmond M. Ocular leprosy. ClinDermatol. 2015; 33(1): 79-89.
[2]. Courtright P. The epidemiology of ocular complications of leprosy.Indian J Lepr. 1998; 70(1): 79-89
[3]. Samantha SK, Das D. Recent advances in ocular leprosy. Indian J Lepr. 2007; 79(2): 135-50.
[4]. Acharya BP. Clinical observation on iridocyclitis in leprosy patients. Ind J Ophthalmol. 1982; 30(2): 65-8.
[5]. Espiritu CG, Gelber R, Ostler HB. Chronic anterior uveitis in leprosy: an insidious cause of blindness. Br J Ophthalmol. 1991; 75(5): 273-5.
[6]. Suryawanshi N. Clinical manifestations of iridocyclitis in leprosy. Indian J Lepr. 1985; 57(3): 549-55.
[7]. Courtright P, Lewallen S, Tungpakom N, Cho BH, Lim YK, Lee HJ, Kim SH. Cataract in leprosy patients: cataract surgical coverage, barriers to acceptance of surgery and outcome of surgery in a population based survey in Korea. Br J Ophthalmol; 2001; 85(6): 643-7.
[8]. Rathinam S, Prajna L. Hypopyon in leprosy patients. J Postgrad Med. 2007; 53(1): 46- 47.
[9]. MacLean H, McKelvie P. Recurrent scleritis in lepromatous leprosy. Aust N Z J Ophthalmol. 1998; 26(1): 51-5.
[10]. Reddy SC, Raju BD. Ocular lesions in the inmates of leprosy rehabilitation centre. Int J of Biomed Sci 2006; 2(3): 289-294.
[11]. Campos WR, Orefice F, Sucena MA, Rodrigues CA. Bilateral iridocyclitis caused by Mycobacterium leprae diagnosed through paracentesis. Indian J Lepr. 1998 Jan-Mar; 70(1):27-31.
[12]. Rathinam SR, Leprosy uveitis in the developing world. IntOphthalmolClin. 2010; 50(2):99-111.
[13].Parikh R, Thomas S, Muliyil J, Parikh S, Thomas R. Ocular manifestation in treated Multibacillary Hansen’s disease. Int J Lepr Other Mycobact Dis; 2002; 70(2): 121-4
[14]. Nepal BP, Shrestha UD. Ocular findings in leprosy patients in Nepal in the era of multidrug therapy.Am J Ophthalmol. 2004; 137(5): 888-92.
[15]. Mahendradas P, Avadhani K, Ramachandran S, Srinivas S, Naik M, Shetty KB. Anterior segment optical coherence tomography findings of iris granulomas in Hansen’s disease: a case report. J Ophthalmic Inflamm Infect. 2013; 3 (1): 36.
[16]. Messmer EM, Raizman MB, Foster CS. Lepromatous uveitis diagnosed by iris biopsy. Graefes Arch ClinExpOphthalmol. 1998; 236(9): 717-9.
[17]. Citirik M, Batman C, Aslan O, Adabag A, Ozalp S, Zilelioglu O. Lepromatousiridocyclitis. OculImmunolInflamm. 2005; 13(1): 95-9.
[18]. Reddy SC, Raju BD. Ocular lesions in the inmates of leprosy rehabilitation centre. Int J Biomed Sci. 2006; 2(3):289-94.
Legends
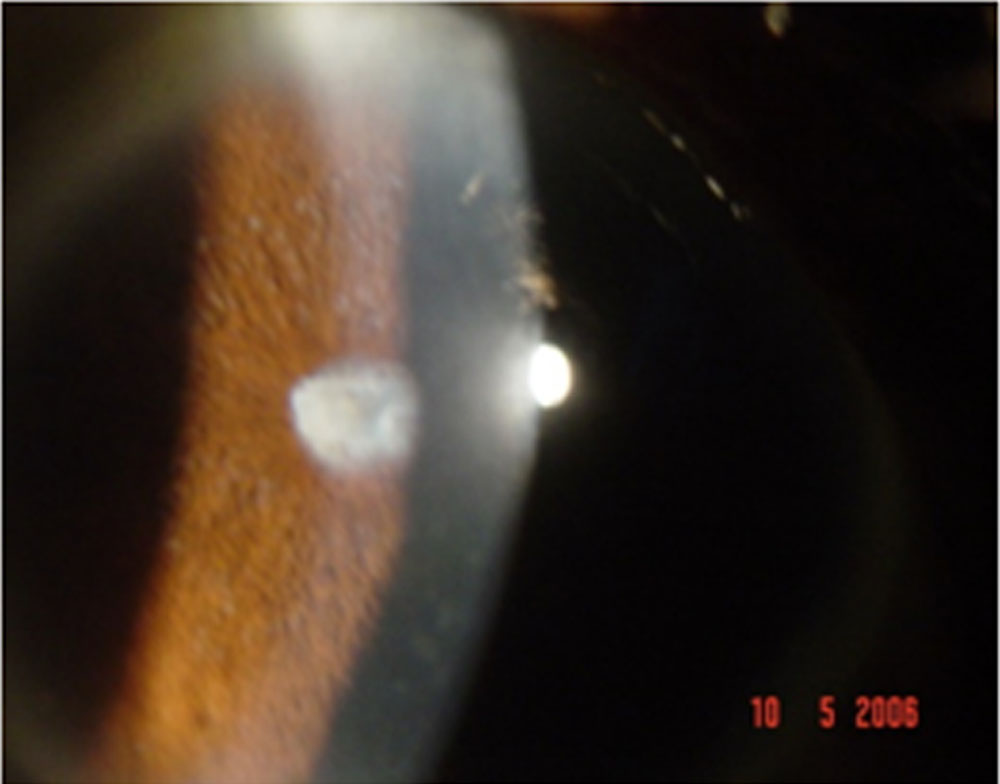
- Figure 1: Mutton fat keratic precipitates and iris pearls
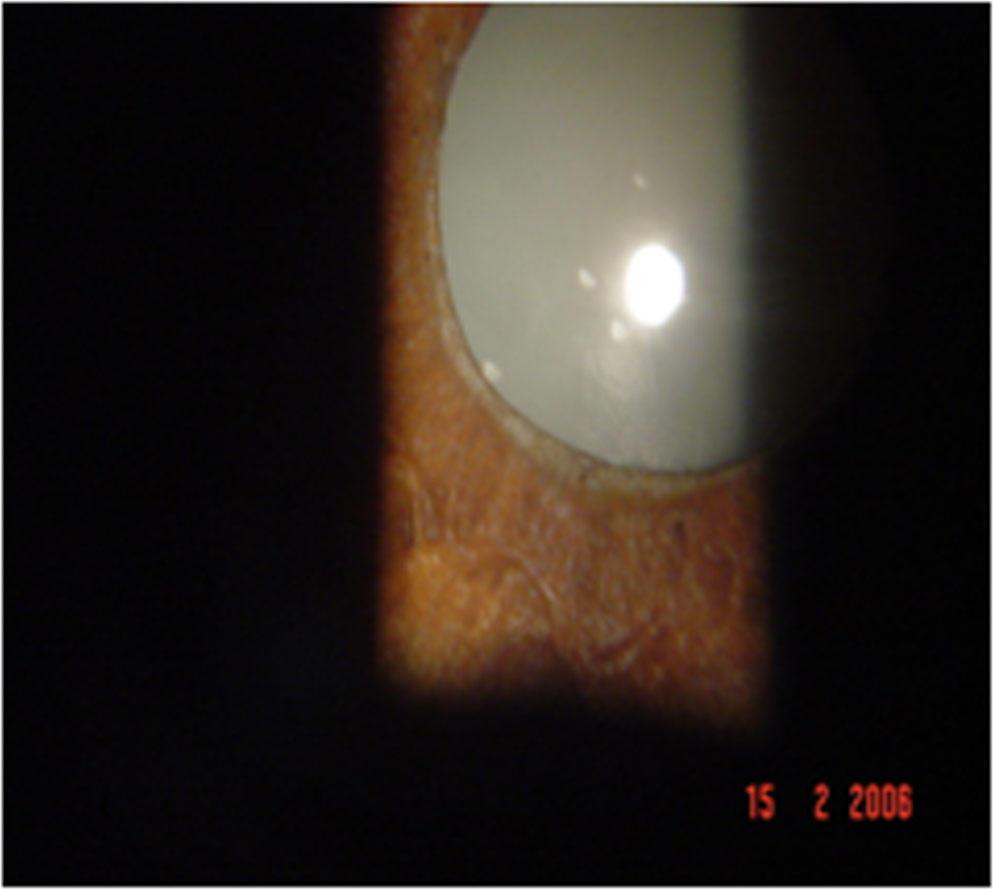
- Figure 2: Complicated cataract in chronic uveitis
- Figure 3: Iris sphincter atrophy with dilated pupil
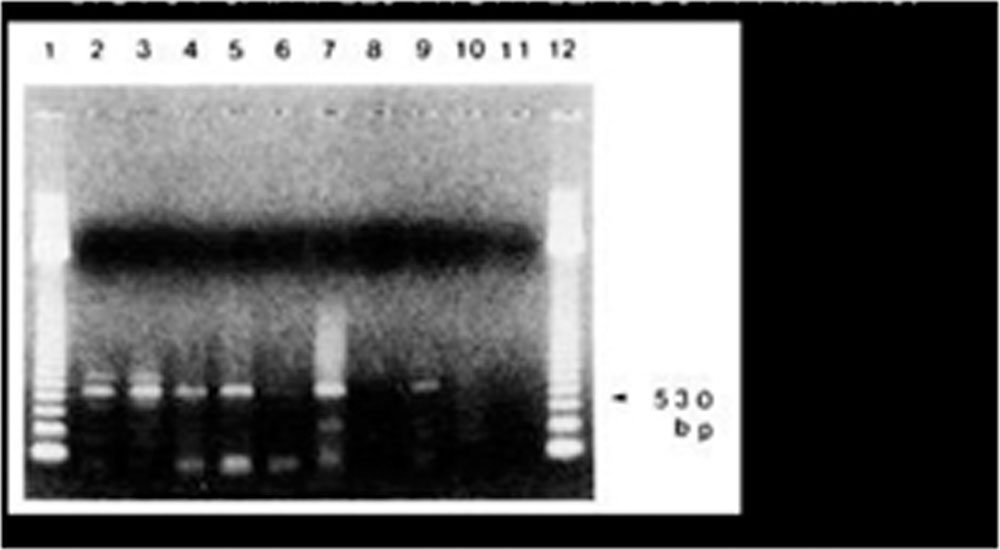
- Figure 4: PCR detection of M. leprae DNA from aqueous humor samples
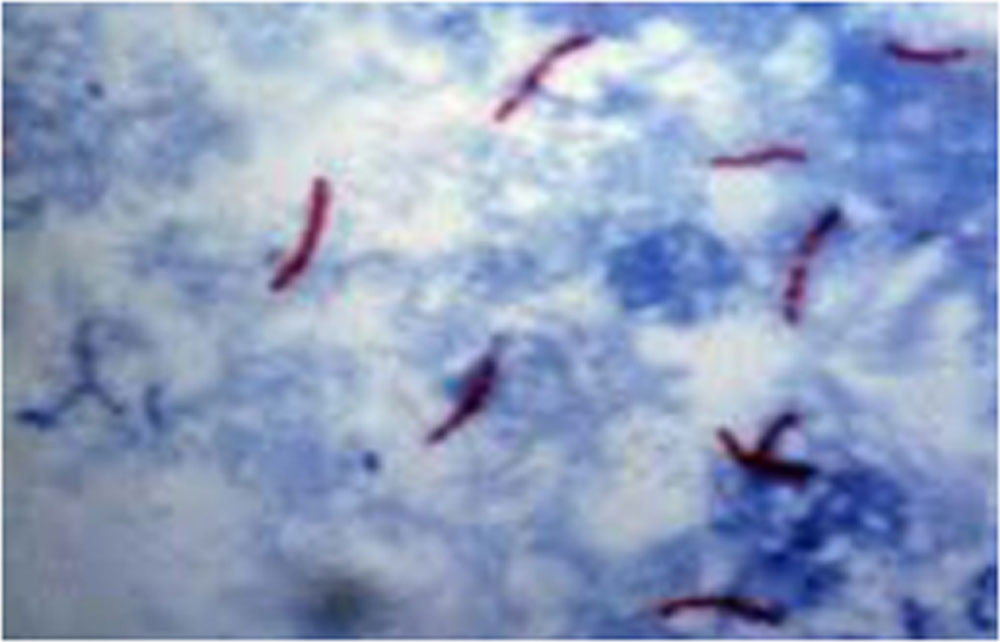
- Figure 5: Smear made from anterior chamber aspirate shows granular beaded dead bacilli and fully formed live bacilli coexisting in the same patient. Zeihl- Neelson x 790
Figure 1: Mutton fat keratic precipitates and iris pearls
Figure 2: Complicated cataract in chronic uveitis
Figure 3: Iris sphincter atrophy with dilated pupil
Figure 4: RT- PCR detection of M. leprae DNA from aqueous humor samples
Figure 5: Smear made from anterior chamber aspirate shows granular beaded dead bacilli and fully formed live bacilli coexisting in the same patient. Zeihl- Neelson x 790




Leave a Comment