Dr. Jitendra Nenumal Jethani, J09433, Dr. Paaraj Dave
![]()
Abstract
Atropine eye drops prevent myopic progression. We did a study to compare the effects of low concentration atropine eye drops (0.01%) with placebo eye drops in children between 5-12 years of age.
All the children taken in the study has history of axial myopic progression. This was a randomised control trial and the subjects were assigned randomly in the case or control group. All the observers were masked to the drops. All investigations were done at 6 months, 1, 2 and 3 years
Atropine eye drops (0.01%) wasprepared by diluting it with lubricating eye drops. The difference in axial growth in the atropine user group in right eye was 0.8 and in left eye was 0.7 mm whereas in the control group the axial growth was 1.6 mm in the right eye and in the left eye is 1.8 mm. The axial growth is halved with usage of atropine eye drops 0.01%
Introduction
Atropine (1%) eye drops isknown to prevent progression of myopia. [1] However, practical problems especially photophobia and accommodation loss are some of the important problems which prevents its widespread use. Since atropine (0.01%) eye drops hasshown promise in retarding the progression of myopia [2-5] we did a study to compare it with placebo drops to see its effect on progression of myopia
Materials and methods
A total of 60 children were included in the study. The children were randomly assigned to each group depending on the computer generated chart for randomisation. All the children had a history of myopic progression of at least -0.75/ year. All children between 5-12 years were included in the study. The Ethical committee approval was taken from the Institutional Ethical Committee of Dr.Thakorbhai V Patel Eye Institute, Vadodara
The atropine 1% eye drops (IntasPharma) wasmixed with Genteal eye drops (Alcon Lab) and atropine 0.01% eye drops wasreconstituted by taking 0.1 ml of Atropine (1%) eye drops and injecting it in 10 ml of Genteal eye drops. The parents were informed to put these drops regularly once a day in the evening.
Cycloplegic refraction, Autorefraction and post mydriatic test with duo chrome testing was done for all the children with axial length measurement (Sonomed Inc.) at 1 month, 3 months, 1 year, 2 and 3 year. Children who did not put drops regularly were excluded. Placebo drops of the same lubricant eye drops were provided to the patients. The measurements were doneby one of the authors who was masked (SS) and the group was assigned by another author (MT)
All the data was recorded in an excel sheet and was analysed using paired t test.
Observation
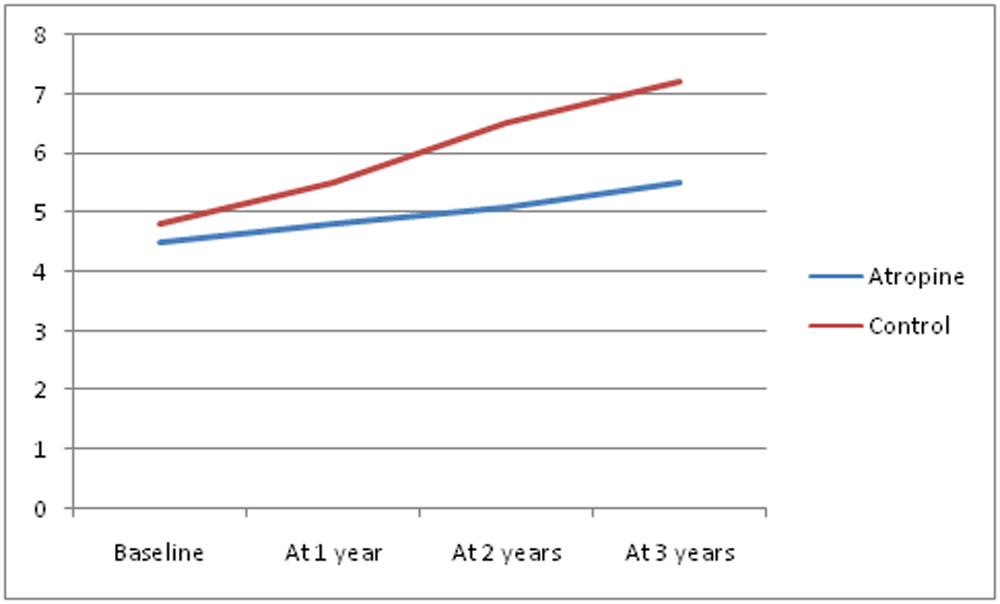
The mean age of the atropine group was 7.2+/- 3.8 years and the control group was 7.2 +/- 5.1 years. The baseline mean spherical equivalent of the spectacle power in atropine group prior to starting the study was -4.5 +/- 3.9 D and in the control group was -4.8 +/- 3.1 D and the baseline axial length in the atropine group was 24.17 +/- 0.9 mm and in the control group was 24.26 +/- 0.8 mm as measured by contact A scan (sonomed Inc.) method
Table 1
| Baseline | At 1 year | At 2 years | At 3 years | |||||
| Atropine | Control | Atropine | Control | Atropine | Control | Atropine | Control | |
| Axial Length | 24.17 +/- 0.8 | 24.26 +/- 0.9 | 24.42 +/- 1.1 | 24.73 +/- 1.2 | 24.61 +/- 0.9 | 25.33 +/- 1.3 | 24.96 +/- 1.1 | 25.77 +/- 1.6 |
| Spherical Equivalent | -4.5 +/- 3.9 | -4.8 +/- 3.1 | -4.8 +/- 4.2 | -5.5 +/- 3.5 | -5.1 +/- 3.8 | -6.5 +/- 4.3 | -5.5 +/- 4.1 | -7.2 +/- 4.6 |
Discussion
The role of atropine eye drops in preventing the progression of myopia has been established. [1-5] ATOM 2 study by Chia et al has shown the role of low concentration atropine eye drops in Singapore children. Our study was done on the Indian children. The drops were reconstituted as these drops were not available in the Indian market. Several studies with smaller followups have also been done showing the role of low concentration atropine in reducing the progression of myopia.
We had a long followup of 3 years and children of Indian origin. None of the previous study has Indian population. Only Chia et al [5] have a longer followup and a larger sample size. Our mean progression both in the control and the atropine eye drops was more than reported by Chia at al [1-5] but was similar to reported by Shu Yi et al [7]
Chia et al [1] in their study ATOM 1 reported a mean increase of axial length of 0.38 mm whereas Shu et al [7] reported an increase in axial length of 0.32 mm in one year.
Conclusion
Atropine 0.01% eye drops can be reconstituted and is well tolerated. The axial length increase is reduced and the myopic progression is also effectively reduced by using these drops in children between 5-12 years of age
References
- Chua WH, Balakrishnan V, Chan YH, Tong L, Ling Y, Quah BL, Tan D. Atropine for the treatment of childhood myopia. Ophthalmology 2006;113:2285-91.
- Chia A, Chua WH, Cheung YB, Wong WL, Lingham A, Fong A, Tan D. Atropine for the treatment of childhood myopia: safety and efficacy of 0.5%, 0.1%, and 0.01% doses (Atropine for the Treatment of Myopia 2). Ophthalmology 2012;119:347-54.
- Chia A, Chua WH, Wen L, Fong A, Goon YY, Tan D. Atropine for the treatment of childhood myopia: changes after stopping atropine 0.01%, 0.1% and 0.5%. Am J Ophthalmol 2014;157:451-457.
- Tan D, Tay SA, Loh KL, Chia A. Topical Atropine in the Control of Myopia. Asia Pac J Ophthalmol (Phila) 2016;5:424-428.
- Cooper J, Eisenberg N, Schulman E, Wang FM. Maximum atropine dose without clinical signs or symptoms. Optom Vis Sci 2013;90:1467-72.
- Chia A, Lu QS, Tan D. Five-Year Clinical Trial on Atropine for the Treatment of Myopia 2: Myopia Control with Atropine 0.01% Eyedrops. Ophthalmology 2016;123:391-9.
- Shu Yi,Yuanshuai Huang, Shi-Zhi Yu, Xi-Jia Chen, Hong Yi and Xiao-Li Zeng. Therapeutic effect of atropine 1% in children with low myopia. J AAPOS 2015;19:426-429.



Leave a Comment