![]()
Dr. Rakesh Juneja, J15026, Dr. Navneet Mehrotra, Dr. Manish Nagpal
Abstract:
Aim: To co-relate angiographic findings of IPCV(Idiopathic Polypoidal Choroidal Vasculopathy) on ICGA with morphological features on OCTA, pre & post treatment
Settings and Design: Single-center, prospective, observational study
Methods and Material:20 eyes of 20 patients, treated with focal laser & anti-VEGF, underwent baseline OCTA and ICGA. All cases were followed up at 1, 3 & 6 months
Statistical analysis used: Descriptive analysis
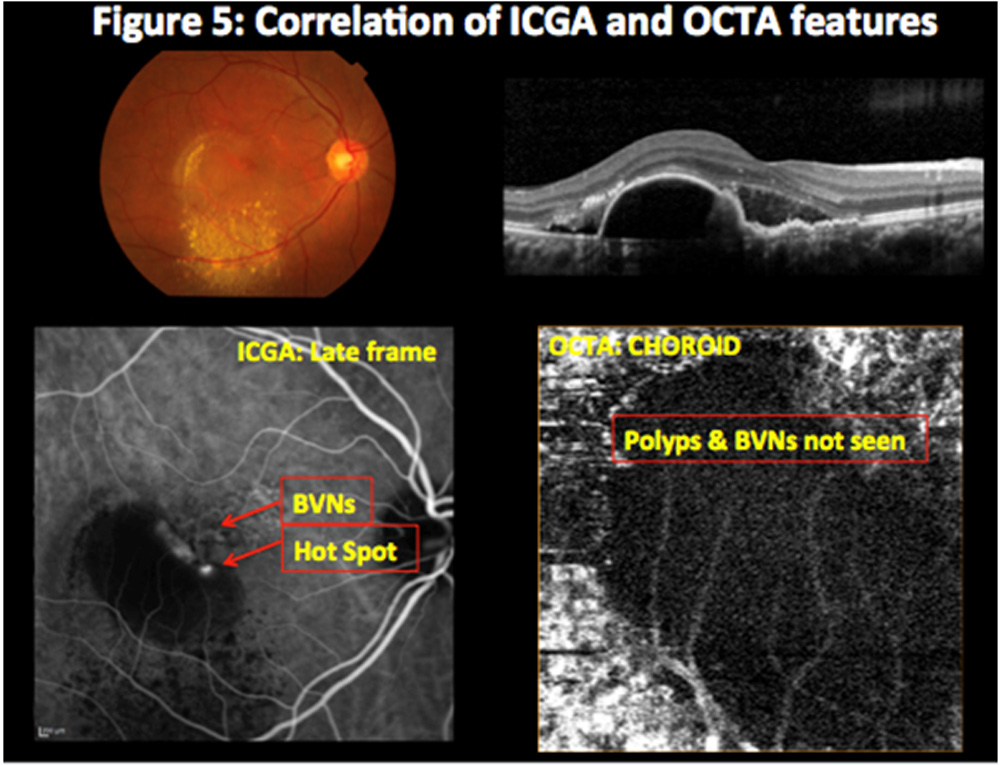
Results:ICGA detected polyps in 20 & BVNs in 17 eyes. OCTA detected each in 14 eyes. Pre treatment, BVN appeared as hyper-flow lesion, polyp as hyper-flow round structure surrounded by a hypo-intense halo corresponding to the hyperfluorescence area, surrounded by hypofluorescence halo and a hyperfluorescent ‘hot spot’ on ICGA. Post treatment revealed decrease in size & number that corresponded to reduced hypofluorescence halo &hyperfluorescent ‘hot spot’ on ICGA
Conclusion: OCTA as a non-invasive tool correlates accurately with ICGA findings for assessing the polyps & BVNs in IPCV & it can be used as an alternative diagnostic test
Keywords: idiopathic polypoidal choroidal vasculopathy, optical coherence tomography angiography, indocyanine green angiography, branch vascular networks, polypoidal lesions
Key Messages:OCTA is a promising “non-invasive” tool that can be used as an “alternative” diagnostic test and is certainly the ‘future of IPCV imaging’Introduction:
IPCV is an acquired, abnormal choroidal vasculopathy.[1,2]IPCV is characterized by polypoidal dilations and choroidal branching vascular networks observed onICGA. [1,2,3,4]OCT shows polypoidal dilations, which are characterized by dome-like elevations of retinal pigment epithelium.[5,6,7] Branching vascular networks appear as ‘double layer sign’. [8]
OCTA works on the principle of ‘decorrelation’ and generates 3-D maps of microvasculature flow pattern from retinal capillary plexus to choroidal vasculature. [9,10]
With the objectives, toassess IPCV features on OCTA and to correlate with ICGA pre & post treatment, a prospective study was conducted, among Indian subset of population, in a tertiary care clinical setting.
Subjects and Methods:
Study design and patient enrolment:
This was a single-center, prospective, observational study with data collected from patients with IPCV diagnosed on OCT and ICGA and underwent OCTA examination at baseline and subsequent follow-ups. All cases that were enrolled provided informed consent to be enrolled in study. The synopsis was reviewed andapproved by the Ethical Committee. Patients recruited through the retina clinic from July 2016 to August 2017, were enrolled in the study, and were assigned a registration number, and no patient-identifiable information was collected. The study adhered to the tenets of the Declaration of Helsinki.
Eligibility criteria and data collection
Data was collected under the following headings: demographic details, history of presenting illness, eye laterality, history and duration of diabetes, hypertension or any other associated systemic co-morbidities, diagnosis of retinal pathology, clinical examination details including visual acuity at presentation and follow-up with imaging (color fundus photography, OCT, OCTA, FA and ICGA), treatment details, complications (if any) and treatment given for that complication. Inclusion criteria were: (1) Cases diagnosed as IPCV on the basis of ICGA and OCT features who underwent OCTA imaging (2) complete availability of documentation till the last follow-up. If any one of the above-mentioned inclusion criteria was not met, patients were excluded from the study.
Data analysis and statistical methods
Various features of IPCV cases at baseline and all follow-ups during the course of study were tabulated and computed using descriptive statistics.
Results:
Baseline Demographics:
Thirty-two eyes of 32 patients diagnosed as IPCV were studied. Out of which, 20 eyes of 20 patients who met our inclusion criteria were included in the study and rest were excluded. The average age of patients in our study group was 54-76 years. Fourteen patients were males and 6 patients were females. All eyes had unilateral presentation. Hypertension was the major co-morbid condition comprising 14 cases (Table 1).
OCTA features of IPCV and its correlation with ICGA:
ICGA detected polyps in 20 & branch vascular networks (BVNs) in 17 eyes. OCTA detected each in 14 eyes. Pre treatment, BVN appeared as hyper-flow lesion, polyp as hyper-flow round structure surrounded by a hypo-intense halo corresponding to the hyperfluorescence area, surrounded by hypofluorescence halo and a hyperfluorescent ‘hot spot’ on ICGA. Post treatment revealed decrease in size & number that corresponded to reduced hypofluorescence halo &hyperfluorescent ‘hot spot’ on ICGA (Figure 1,2,3,4,5). At baseline, OCTA showed 70% sensitivity as compared to ICGA in detecting IPCV (Table 2) whereas during all follow-ups post treatment OCTA revealed 100% sensitivity as compared to ICGA. (Table 3)
Discussion:
Polypoidal lesions in IPCV characteristically appear as hyper-flow round structures on OCTA. However, in certain cases, hypo-flow structures are also observed. This absence of signal does not mean that there is no blood flow; rather, it indicates that blood flow is not within the detection limit of the OCTA device. This could be due to either increased or decreased flow in the polyps and subsequent nonvisualization of the vascular structure. Although choroidal blood flow is known to be higher than retinal blood flow,[11] and some studies of IPCV hemodynamics have suggested that these lesions originate from choroidal vascularization, [12] this hypothesis is very unlikely. High blood flow in the polyps is theoretically possible, but ICGA has revealed that the polyps do not fill very rapidly during early-phase angiography. We hypothesize that the apparent absence of OCTA signal within polypoidal lesions could be due to either the presence of turbulent blood flow inside of the polyps – impeding the representation of this flow – or to the fact that blood circulates only at the periphery of the aneurysmal dilation. The last hypothesis is sustained by the fact that the pigmentary epithelium detachment associated with the polypoidal structure also demonstrates an attenuated OCTA signal.
OCTA allows for the visualization of retinal microvasculature by detecting intravascular linear blood flow, [13,14] and indeed, in our patients, the branching vascular network, which is characterized by linear blood flow, was clearly detected using the OCTA’s priniciple of decorrelation.
In conclusion, the results of our study provide new insight to image IPCVs non-invasively. However, ICGA still stands as the age-old, time-tested, gold-standard modality for diagnosing IPCVs with 100% accuracy at both baseline as well as at all follow-ups. OCTA non-invasively does complement and correlates with ICGA in detecting IPCVs with a fairly good sensitivity of 70% at baseline and equivalent 100% sensitivity at all follow-ups.
OCTA is a noninvasive imaging modality, and we have demonstrated that it allows for the visualization of different structures in IPCV. The branching vascular networks are clearly and consistently visualized as hyper-flow lesions, but further improvements in OCTA knowledge are needed to gather information on the specificity of the different intensity characteristics of polypoidal lesions.
The major limitations of our study are the small sample size and lack of swept source OCTA. The major strength of the study is that it provides data on Indian subset of population, is only the second study in literature incorporating such a large number of IPCV cases and provides data both at baseline as well as on follow-ups.
OCTA thus is a promising non-invasive tool andcorrelates with the information provided by invasive ICGA and with further upcoming advances in bio-medical engineering it seems to be the future of imaging in IPCV.
Abbreviations:
OCT optical coherence tomography
BVN branch vascular network
IPCV idiopathic polypoidal choroidal vasculopathy
OCTA optical coherence tomography angiography
ICGA indocyanine green angiography
References:
- Yannuzzi LA, Sorenson J, Spaide RF, et al: Idiopathic polypoidal choroidal vasculopathy (IPCV). Retina (Philadelphia, Pa.) 1990;10:1–8.
- Laude A, Cackett PD, Vithana EN, et al: Polypoidal choroidal vasculopathy and neovascular age-related macular degeneration: same or different disease? ProgRetin Eye Res 2010;29:19–29.
- Ciardella AP, Donsoff IM, Huang SJ, et al: Polypoidal choroidal vasculopathy. SurvOphthalmol 2004;49:25–37.
- Spaide RF, Yannuzzi LA, Slakter JS, et al: Indocyanine green videoangiography of idiopathic polypoidal choroidal vasculopathy. Retina (Philadelphia, Pa.) 1995; 15:100–110.
- Sa H-S, Cho HY, Kang SW: Optical coherence tomography of idiopathic polypoidal choroidal vasculopathy. Korean J Ophthalmol 2005;19:275–280.
- Iijima H, Imai M, Gohdo T, et al: Optical coherence tomography of idiopathic polypoidal choroidal vasculopathy. Am J Ophthalmol 1999;127:301–305.
- Otsuji T, Takahashi K, Fukushima I, et al: Optical coherence tomographic findings of idiopathic polypoidal choroidal vasculopathy. Ophthalmic Surg Lasers 2000;31:210–214.
- Sato T, Kishi S, Watanabe G, et al: Tomographic features of branching vascular networks in polypoidal choroidal vasculopathy. Retina (Philadelphia, Pa.) 2007;27:589–594.
- Nagpal M, Juneja R. Panoramic Imaging With OCTA. Retina Today. 2017 April
- Nagpal M, Singh SS. OCT angiography in retinal and choroidal diseases. Retina Today. 2016;11(8):57-64.
- Muir ER, Duong TQ: MRI of retinal and choroidal blood flow with laminar resolution. NMR Biomed 2011;24:216–223.
- Watanabe G, Fujii H, Kishi S: Imaging of choroidal hemodynamics in eyes with polypoidal choroidal vasculopathy using laser speckle phenomenon. Jpn J Ophthalmol 2008;52:175–181.
- Jia Y, Tan O, Tokayer J, et al: Split-spectrum amplitude-decorrelation angiography with optical coherence tomography. Opt Express 2012;20:4710–4725.
- Jia Y, Bailey ST, Wilson DJ, et al: Quantitative optical coherence tomography angiography of choroidal neovascularization in age-related macular degeneration. Ophthalmology 2014;121:1435– 1444.
Table no. 1: Baseline Demographics
| PARAMETERS | n (%) |
| No. of patients | 20 |
| No. of eyes | 20 |
| Female | 14(70%) |
| Male | 6(30%) |
| Age (mean) | 65 |
| Age (Range) | 54-76 |
| Unilateral | 20(100%) |
| Bilateral | 0 |
| Hypertension | 14 |
Table no 2: Comparison of sensitivity between OCTA and ICGA to screen IPCV at
baseline
| TOOL | PROPERTY | No. of IPCV
cases
(at Baseline) |
No. of
patients diagnosed (at baseline) |
Sensitivity
to screen at baseline |
|
ICGA |
INVASIVE |
20 |
20 |
100% |
|
OCTA |
NON INVASIVE |
20 |
14 |
70% |
Table no. 3: Comparison of sensitivity between OCTA and ICGA to screen IPCV at
Follow-ups
| TOOL | PROPERTY | No. of IPCV
cases
(at Follow-up) |
No. of
patients diagnosed (at follow-up) |
Sensitivity
to screen at follow-up |
|
ICGA |
INVASIVE |
6 |
6 |
100% |
|
OCTA |
NON INVASIVE |
6 |
6 |
100% |
Figure 1: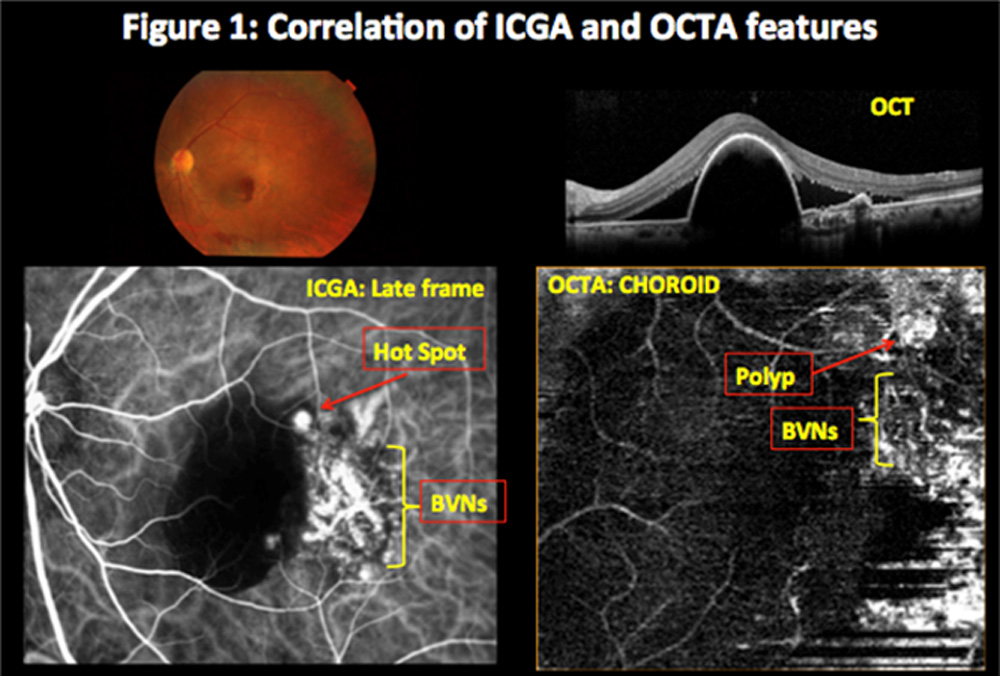
Figure 2:
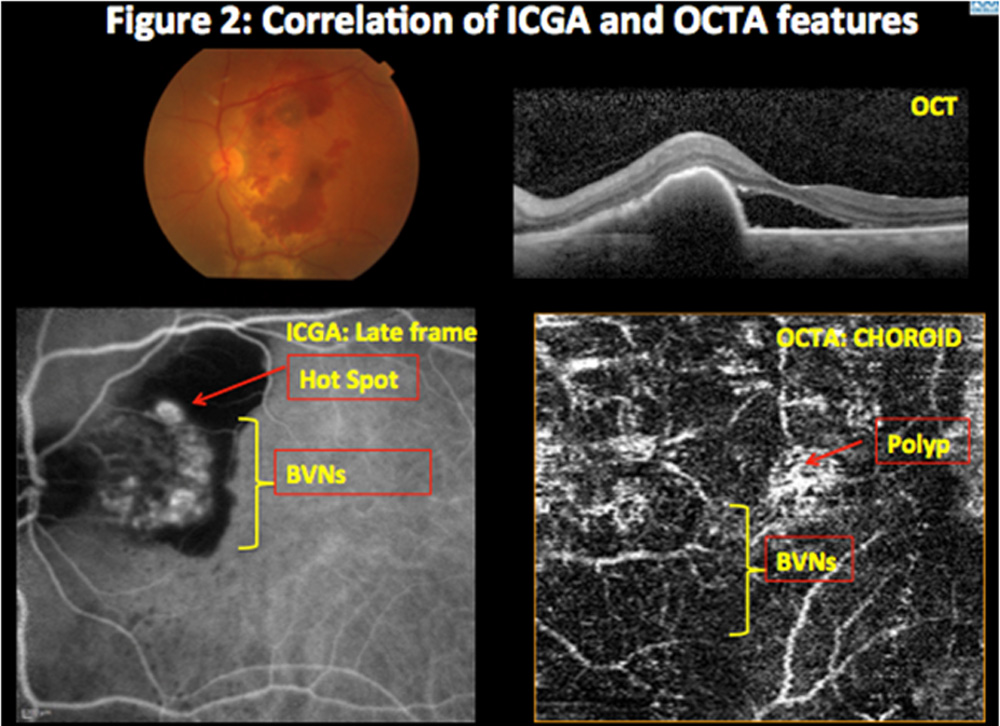
Figure 3:
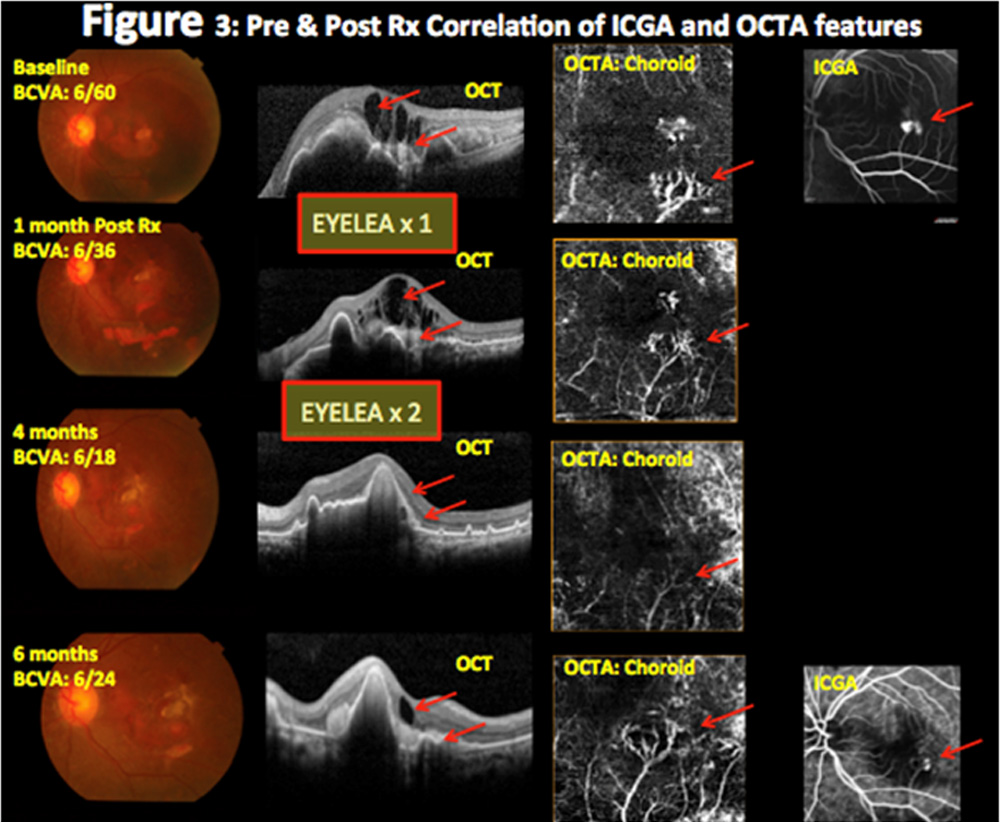
Figure 4: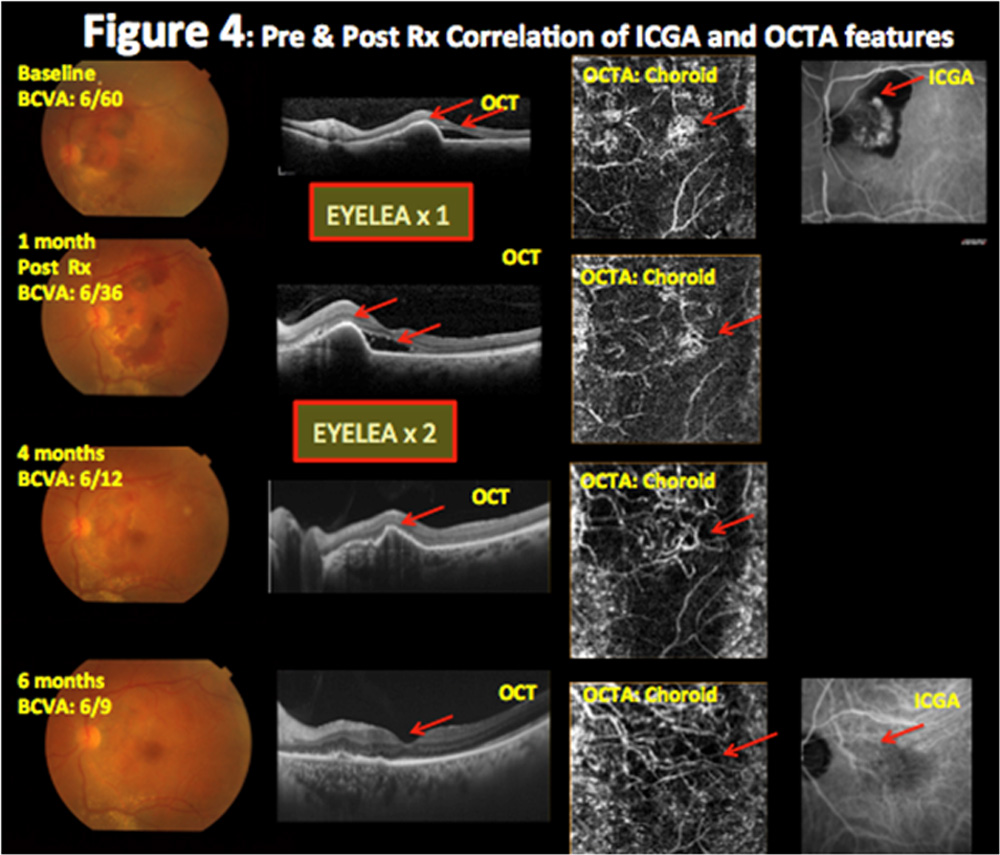
Figure 5:


Leave a Comment