Dr. Parul Goyal (P19355 )
Abstract
Purpose: Todetermine the effect of oral omega 3 fatty acid (w3FA) supplementation in LASIK associated dry eye.
Materials and Methods: In this prospective, placebo controlled, open label study patients undergoing LASIK were randomized to receive either 1.2 gms /day of oral w3FA (treatment group) or 400mg/day Vitamin E (placebo group) for 1 week before and 12 weeks after surgery. Ocular surface disease index (OSDI), tear break up time (TBUT),ocular surface staining and Schirmer scores were evaluated preoperatively and subequently at 1 week , 1 month and 3 months after LASIK. Absolute values as well as mean change from baseline of these outcome measures at each visit was compared between the two groups.
Results
The study included 30 patients in each group. All parameters were comparable at baseline.Mean increase of OSDI from baseline was comparable in both groups. Both groups had significant decrease in TBUT as compared to baseline (P <0.01) but the intergroup difference was not significant (P >0.05) at any post operative visit. Greater number of eyes in placebo group (p=0.009) had some degree of conjunctival staining with Lissamine green dye at 3 months when compared to the treatment group. The Schirmer score at 3 months was significantly higher (p=0.003) in the treatment group (30.52±0.90 mm) than the placebo group (25.73 ± 1.26mm).
Conclusions
Supplementation with w3FA in patients undergoing LASIK , had a positive influence on tear secretion ,while tear film stability remained largely unaffected when compared to the placebo group.
Key Words: Omega -3 fatty acids, LASIK, dry eye.
Introduction
Laser in situ keratomileusis (LASIK) is a safe and effective surgical option for treatment of refractive errors. However upto 95% of patients may experience symptoms of dryness immediately after LASIK 1 which may persist in upto 20 to 40 % of patients even at 6 months.2,3Damage to corneal nerves is one of the key driving factors in causing dry eyes post LASIK.4 In the human cornea, stromal nerves courses anteriorly through the stroma to form the subbasal nerve plexus. The central subbasal and stromal nerve fiber bundles on imaging by confocal microscopy are virtually absent immediately after LASIK because of transection of nerves during flap creation and subsequent stromal photoablation.5The nerves have been shown to regrow after LASIK but even at 5 years their density remains less than the preoperative levels.6 This decreased innervation causes impairment of corneal sensations and consequent disruption of the complex interaction between the afferent sensory nerves of the ocular surface and the efferent autonomic nerves to the lacrimal gland i.e. the lacrimal functional unit.7-9 Also a denervated cornea leads to a drop in the blink rate thus exposing the ocular surface for longer period of time and making it more prone to develop symptoms and signs of dry eyes.3
Surgical injury to epithelium during refractive surgery has also been shown to result in subclinical corneal inflammation and increase in cytokines and interleukins (IL) 6 and 8 in tears , which may be partially responsible for triggering the symptoms and signs of dry eye.10,11LASIK has also been associated with loss of goblet cells secondary to placement of the suction ring or corneal nerve damage, with a subsequent reduction in goblet cell mucin, which is a stabilizing molecule in the tear film. 12
Omega 3 Fatty Acids (w3 FA) are essential fatty acids which cannot be synthesized by the body and need to be obtained by dietary means. These include Alpha Linolenic Acid (ALA), Eicosapentanoic Acid (EPA), and Docosahexanoic Acid (DHA).Injury to the cornea induces an upregulation of nerve growth factor (NGF) secretion .13 Combinations of NGF plus DHA and pigment epithelial derived factor (PEDF) plus DHA have been shown to improve nerve regeneration after injury to cornea 14,15Thus supplementation with DHA containing w3FA formulations may be of particular significance in LASIK.
Increased consumption of w3 FA such as EPA, and DHA results in greater proportions of these accumulating in cell membrane phospholipids at the expense of arachidonic acid (AA). These w3FA components then compete with AA as a substrate for cyclooxygenase (COX) 2 and 5 lipooxygenaze (LOX).16,17The eicosanoids generated from the w3 FA substrates i.e. prostaglandin (PG) E3, thromboxane (TX) A3 and leukotriene (LT) B5 have a predominantly anti-inflammatory action vis a vis the eicosanoids generated from AA (PG E2, TX A2 and LTB4) which are pro inflammatory .18Consumption of w3FA has also been shown to improve goblet cell density in patients of dry eyes.19,20
Potential benefits of w3FA have been documented in dry eyes of varied aetiologies including meibomian gland dysfunction (MGD), computer vision associated dry eye and contact lens induced dry eyes.19-25Consideringthe multiple actions of w3FAwhich may have a positive influence on the pathophysiology of post LASIK dry eye i.e nerve regenerating potential, anti-inflammatory activity and effect on goblet cell density , the present study was prospectively conducted to determine the role of these agents in preventing or reducing the signs and symptoms of dry eye after LASIK.
Materials and Methods
Patients attending the Cornea and Refractive Services of the Advanced Eye Centre, Post Graduate Institute of Medical Education and Research, Chandigarh, India, for undergoing LASIK were prospectively recruited between July 2014 to September 2015. The study was approved by the institute ethics committee and adhered to the tenets of the Declaration of Helsinki. Sixty consecutive patients who were considered fit for the procedure, were allocated either to a treatment group or a placebo group using a random number table, after obtaining a written signed consent.
The inclusion criteria for the study were (1) age 20 years or greater; (2) stable refractive error for last 1 year; (3) maximum manifest refraction spherical equivalent (MRSE) of -6.00 Diopters (D). The exclusion criteria were (1) presence of allergic conjunctivitis, anterior blepharitis or MGD; (2) lacrimal drainage abnormalities e.g. punctual stenosis or nasolacrimal duct obstruction ; (3) previous history of lacrimal gland pathology e.g. dacryoadenitis or lacrimal gland surgery (4) presence of structural or functional lid anomalies; (5) presence of pre-existing dry eye due to any cause, defined for the purpose of the study as Schirmer I test without anaesthesia <10 mm /5 minutes and Ocular Surface Disease Index (OSDI) questionnaire score of ≥ thirteen;26 (6) any condition which made the patient an unsuitable candidate for LASIK e.g.epithelial basement membrane disease or a history of recurrent corneal erosions, severe progressive or unstable myopia, keratoconus, anticipated residual stromal bed thickness less than 300 μm post LASIK ; (7) current use of systemic corticosteroid or immunosuppressive therapy; (8) patients taking anti depressants, anti histamines or anticoagulants (9) presence of autoimmune diseases, collagen vascular diseases, diabetes mellitus; (10)pregnant, nursing, or lactating women; (11) patients having malabsorption syndromes; (12) allergy to fish oils. Contact lenses if being used were stopped at least 2 weeks prior to recruiting the patient for the study.
LASIK procedures were performed by 2 surgeons (CM, AKJ). Flaps for all patients were created using the IntraLase femtosecond [IFS] 150 KHz laser, (Abbot Medical Optics Santa Ana, California [AMO]). To ensure consistency of surgical parameters all flaps were programmed with the following settings: 100μm thickness, 8.5 to 9.0-mm diameter, 55° superior hinge angle, 120° side-cut angle, laser raster patterns, spot/line separation of 7/7μm, and stromal energy of 0.76 microjoules with a side-cutenergy of 1.2 microjoules.The MEL80 excimer laser (Carl Zeiss Meditec AG, Jena, Germany) was used for ablation in all eyes.
Intervention:
Both groups were given a topical antibiotic( moxifloxacin 0.5% 4 times/day for 2 weeks after surgery), topical steroid ( prednisolone acetate 1% 4 times/day for the first week and on tapering dose for next 3 weeks) and an artificial tear substitute containing polyethylene glycol 0.4% and propylene glycol 0.3% (Systane Ultra, Alcon Laboratories) 6 times a day, for a period of 12 weeks after the surgery. The treatment group in addition received oral supplementation of a triglyceride formulation of w3FA, with each capsule containing 300mg of w3 FA derivatives (Capsule OSMEGATM, Allergan containing 180 mg of EPA and 120mg of DHA). The dosing schedule was 2 capsules in the morning and 2 in the evening amounting to a total supplementation of 1.2 gms/day (720 mg of EPA and 480 mg of DHA). The w3FA supplementation was started 1 week prior to surgery and continued for 12 weeks after LASIK as dry eye symptoms have been reported to peak between 1 week and 3 months post surgery. 4 The placebo group received vitamin E capsules 400mg/day (Capsule Evion, Merck Serono Limited, Mumbai,India) for a similar duration.The study design was open label with the patients of both groups being aware of the treatment received.
Clinical examination:
Patients were examined in detail at baseline (i.e. prior to staring the w3FA oral supplementation or Vitamin E Capsules) and at 1week, 1month and 3 months after undergoing LASIK. At each visitprior to starting clinical examination patients were required to fill the Ocular Surface Disease Index (OSDI) questionnaire, which contains 12 questionsand is a validated tool to assess dry eye symptoms. The overall OSDI score defines the ocular surface as normal (0-12 points), or as having mild (13-22 points), moderate (23-32 points) or severe (33 -100 points) disease i.e. increasing scores on the OSDI questionnaire represent a greater degree of disability. 26
Examination at each visit included in addition to a routine clinical observations, measurement of tear break up time (TBUT) to assess tear film stability , corneal and conjunctival staining using fluorescein and 1% lissamine green dyes respectively, to assess the ocular surface health and Schirmer 1 test (without anaesthesia) to assess total tear secretion (basic and reflex) . All examinations were done by a single observer who was not masked to the intervention received by the patients.
Tear break up time (TBUT) was measured first using the Lowther technique.27 Briefly a sterile fluorescein strip was dampened with a drop of non-preserved saline solution, and the strip was touched to the superior palpebral conjunctiva. Patients were asked to blink several times for even distribution of the fluorescein with the tear film. They were then instructed to open their eyes without blinking. The time between the opening of the eyes and the appearance of the first dry spot was measured in seconds 3 times. The average of the 3 measurements was recorded as the final TBUT.
Corneal and conjunctival staining was assessed next using fluorescein and 1% lissamine green dyes respectively. The staining pattern was assessed and scored using the standardized methods recommended by the National Eye Institute/Industry workshop.28 In this grading system, the cornea is divided into five areas for each eye i.e. central, inferior, nasal, temporal and superior. The amount of staining in each area is graded from 0 to 3 according to intensity of fluorescein staining with a maximum staining score of 15. Values above 3 are considered abnormal. Conjunctival staining with Lissaminegreen is graded on a scale from 0 to 3 in three areas each (inferior paralimbal, superior paralimbal, and peripheral) of the nasal and temporal conjunctiva with a maximum score of18 for each eye and values above 3 being considered abnormal.
Tear secretion was assessed by the Schirmer 1 test (without anaesthesia) using Whatman filter no.41 strips measuring 5 X35 mm which were placed in lower fornix at junction of lateral and middle third of lower lid. After 5 minutes the strips were removed and a measurement (in millimeters) of the wet area of the strip was made. The Schirmer test was done,with a minimum gap of at least 1 hour after the ocular staining measurements, in order to let the tear film stabilize.
Outcome measures: Primary outcome measures were mean change in tear secretion (Schirmers score in mm) and tear film stability (TBUT in seconds) from baseline to 3 months. Secondary outcome measures were change in ocular surface staining and OSDI questionnaire score from baseline to 3 months.
Statistical analysis: Normality of the measurable data was assessed using Kolmogorov-Smirnov Test. For normally distributed measurable data, Student’s t-test (un-paired) was applied to compare means of the two groups, while for skewed data Mann-Whitney U Test was applied. Categorical data was expressed as frequencies and percentages. The association of various categorical/classified data within the two groups was analysed using Chi-square Test or Fisher’s exact test. For parameters observed over different points of time, the trend was observed/ compared using repeated measures analysis of variance. Both within group and inter group comparisons were made and change from baseline was compared for each variable evaluated. A ‘P’ value (two sided) of <0.05 was considered to be significant in all the tests. Calculations were performed using SPSS (version 22.0; SPSS Inc., Chicago, IL, USA).
Results: The study comprised 60 participants who were divided into 2 groups: a treatment group consisting of 60 eyes of 30 patients and a placebo group consisting of 59 eyes of 30 patients (one patient underwent unilateral LASIK). Both groups were age matched (p= 1.0) with mean age being 23.6 ± 2.4 years (range, 20 – 32 years) in the treatment group and 23.6 ±3.4 years (range, 20 -34 years) in the placebo group. Of the 60 patients 27(45%) were males and 33 (55%) were females. Mean ablation depth in the treatment group (79.03 ± 14.18µm) was comparable to that of the placebo group ( 77.58 ± 13.22 µm).Table 1 shows various parameters (OSDI, TBUT, Schirmer, corneal fluorescein staining and conjunctival staining with Lissamine green dye) at baseline, which were comparable between the two groups.
| Table 1: Baseline Comparison of Clinical Measures Between the Treatment Group and Placebo Group | |||
| Parameter | Treatment Group
Mean ± SEM |
Placebo Group
Mean ± SEM |
P |
| OSDI | 0.98 ± 0.29 | 0.72 ± 0.23 | 0.491 |
| Schirmer (mm) | 25 ± 1.17 | 27.49 ± 1.06 | 0.119 |
| TBUT (seconds) | 10.15 ±0.29 | 10.78 ± 0.53 | 0.299 |
| Corneal fluorescein staining scores(%eyes) | |||
| 0 | 98.3 | 94.9 | 0.364 |
| 1 | 1.7 | 5.1 | |
| Conjunctival LissamineGreen staining scores (% eyes) | |||
| 0 | 98.3 | 98.3 | 1.0 |
| 1 | 1.7 | 1.7 | |
| SEM, standard error of mean. | |||
| Table 2: Comparison of absolute OSDI scores Between the Treatment Group and Placebo Group | |||
| Parameter | Treatment Group
Mean ± SEM |
Placebo Group
Mean ± SEM |
P |
| OSDI Baseline | 0.98 ± 0.29 | 0.72 ± 0.23 | 0.491 |
| OSDI 1week postop. | 5.15 ± 0.63 | 4.75 ± 0.65 | 0.66 |
| OSDI 1 month postop. | 4.78 ± 0.67 | 5.34 ± 0.79 | 0.587 |
| OSDI 3 months postop. | 2.92 ± 0.58 | 3.49 ± 0.47 | 0.453 |
| OSDI, ocular surface disease index; Post op., postoperative; SEM, standard error of mean | |||
OSDI scores: The absolute OSDI score was comparable at baseline and at all subsequent visits in both groups (Table 2).
Mean change in OSDI scores from baseline was also calculated and within group and inter group comparisons were made (Table 3).
| Table 3: Inter group comparison of mean change in OSDI scores from baseline | |||||
| Group | Mean ± SEM | Intergroup
comparison (P value) |
|||
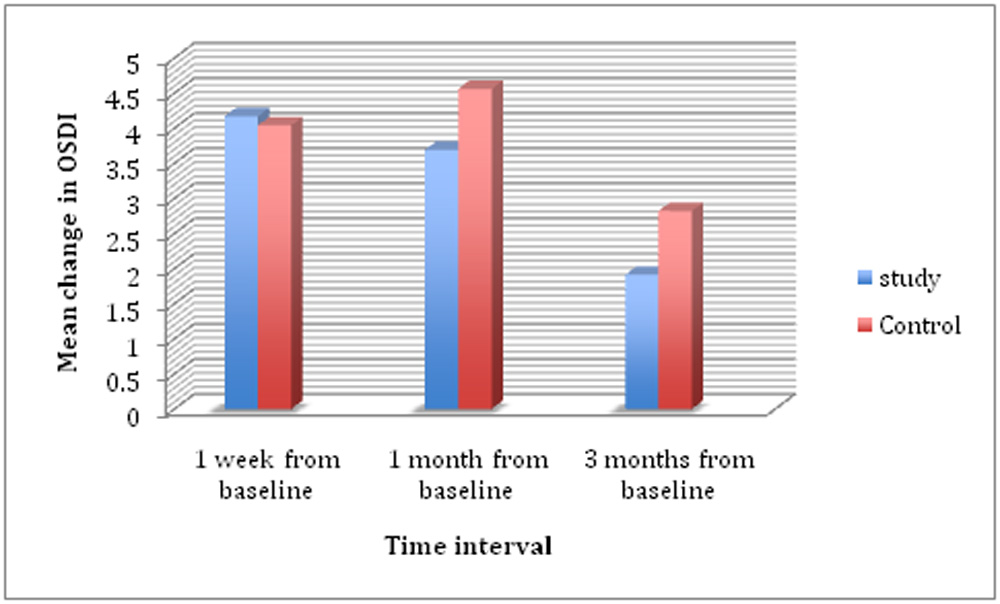
| Mean change at 1 week postop. from baseline | Treatment | 4.17± 0.57 | 0.876 | ||
| Placebo | 4.04 ± 0.65 | ||||
| Mean change at 1 month postop. from baseline | Treatment | 3.69 ± 0.52 | 0.358 | ||
| Placebo | 4.56 ± 0.81 | ||||
| Mean change at 3 months postop. from baseline | Treatment | 1.91 ± 0.60 | 0.267 | ||
| Placebo | 2.82 ± 0.53 | ||||
| OSDI, Ocular Surface Disease Index; SEM , standard error of mean; *P<0.05 | |||||
Within group comparisons showed that for both groups mean change i.e. increase of OSDI was significant at all post operative visits upto 3 months (all P’s <0.01) when compared to baseline . Inter group comparisons however revealed no significant difference between the treatment and placebo group i.e. mean change of OSDI score from baseline was similar in both the groups .
| Table 4: Comparison of TBUT between the Treatment Group and Placebo Group | |||
| Parameter | Treatment Group
Mean ± SEM (seconds) |
Placebo Group
Mean ± SEM (seconds) |
P |
| TBUT Baseline | 10.15 ± 0.29 | 10.78 ± 0.53 | 0.299 |
| TBUT 1week postop. | 7.38 ± 0.37 | 7.44 ± 0.42 | 0.920 |
| TBUT 1 month postop. | 6.93 ± 0.48 | 6.84 ± 0.40 | 0.896 |
| TBUT 3 months postop. | 8.10 ± 0.22 | 7.45 ± 0.44 | 0.181 |
| TBUT, tear break up time; Post op., postoperative; SEM, standard error of mean | |||
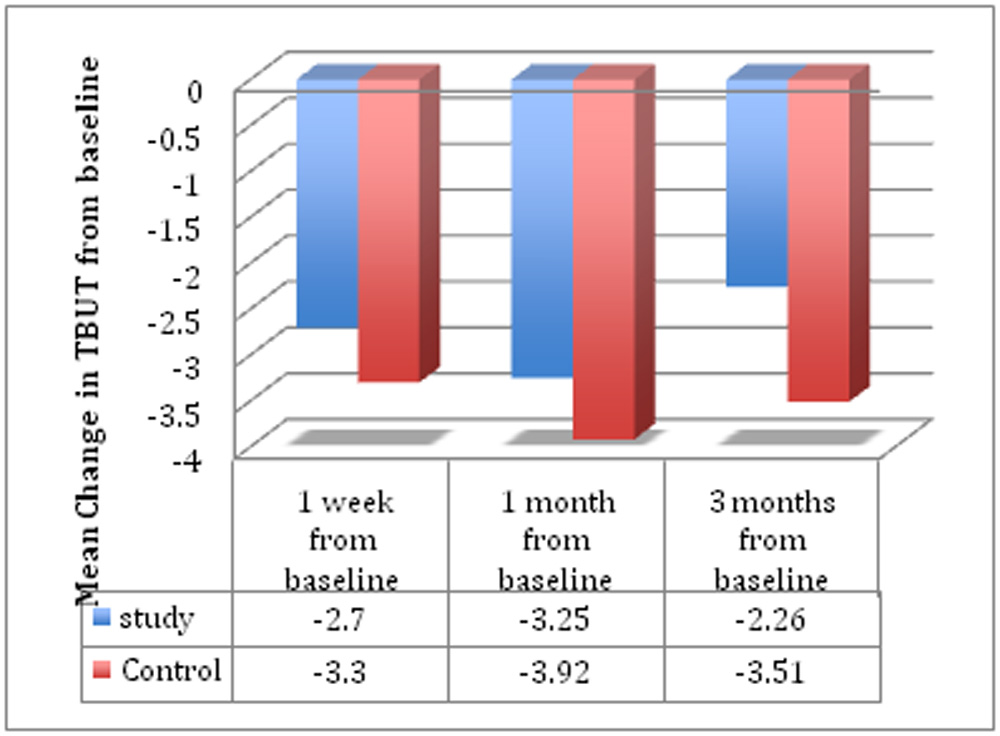
TBUT: The mean TBUT was comparable at baseline and at all subsequent visits in both groups (Table 4). Mean change in TBUT from baseline was also calculated and within group and inter group comparisons made (Table 5). Within group comparison showed that for both groups mean change i.e. decrease of TBUT was significant at all post operative visits (all P’s <0.01) when compared to baseline . However on intergroup analysis the mean change i.e. mean decrease in TBUT from baseline was similar in both groups (all P’s >0.05) at all the postoperative visits .
| Table 5: Inter group comparison of mean change in TBUT from baseline | ||||
| Group | Mean ± SEM
(seconds) |
Intergroup
comparison (P value) |
||
| Mean change at 1 week postop. from baseline | Treatment | -2.7 ± 0.38 | 0.435 | |
| Placebo | -3.3 ± 0.63 | |||
| Mean change at 1 month postop. from baseline | Treatment | -3.25 ± 0.50 | 0.404 | |
| Placebo | -3.92 ± 0.61 | |||
| Mean change at 3 months post op from baseline | Treatment | -2.26 ± 0.35 | 0.105 | |
| Placebo | -3.51± 0.69 | |||
| Postop., postoperative; TBUT, Tear Break Up Time; SEM , standard error of mean; *P<0.05 | ||||
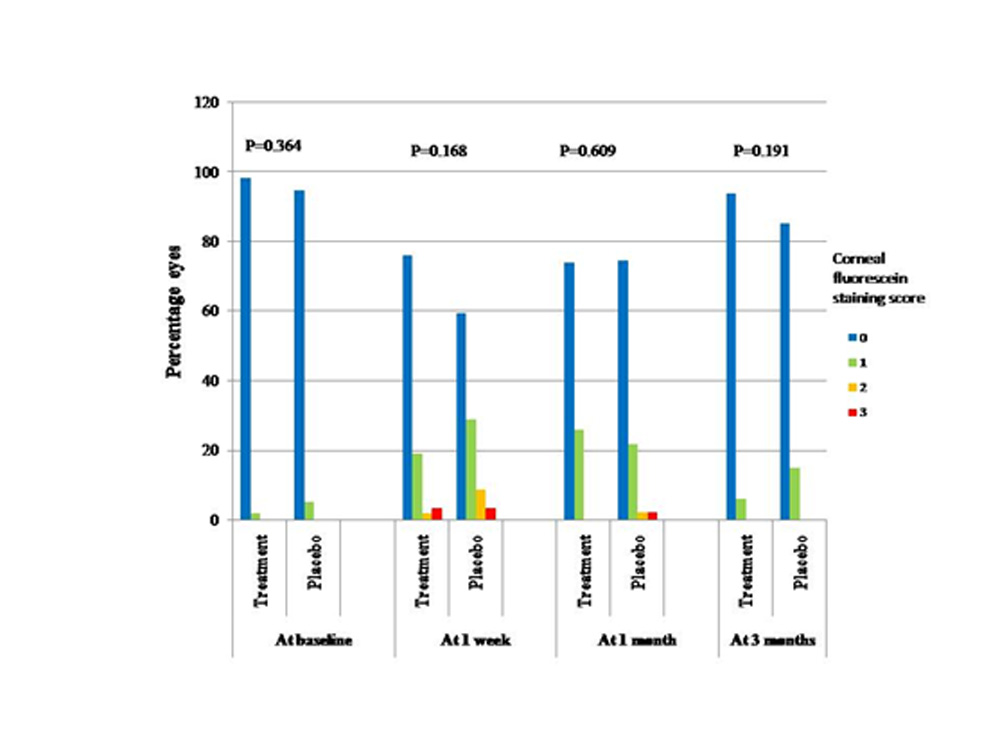
 Ocular Surface Staining: At baseline 98.3% and 94.9% eyes in the treatment and placebo groups respectively had no corneal staining with fluorescein while 1.7% and 5.1% eyes had a corneal staining score of 1 (p=0.364). No eyes in either group had a corneal staining score greater than 3. At all post operative visits upto the 3rd month also no significant difference was seen in the corneal staining scores in either of the twogroups(Figure1).
Ocular Surface Staining: At baseline 98.3% and 94.9% eyes in the treatment and placebo groups respectively had no corneal staining with fluorescein while 1.7% and 5.1% eyes had a corneal staining score of 1 (p=0.364). No eyes in either group had a corneal staining score greater than 3. At all post operative visits upto the 3rd month also no significant difference was seen in the corneal staining scores in either of the twogroups(Figure1).
Figure 1
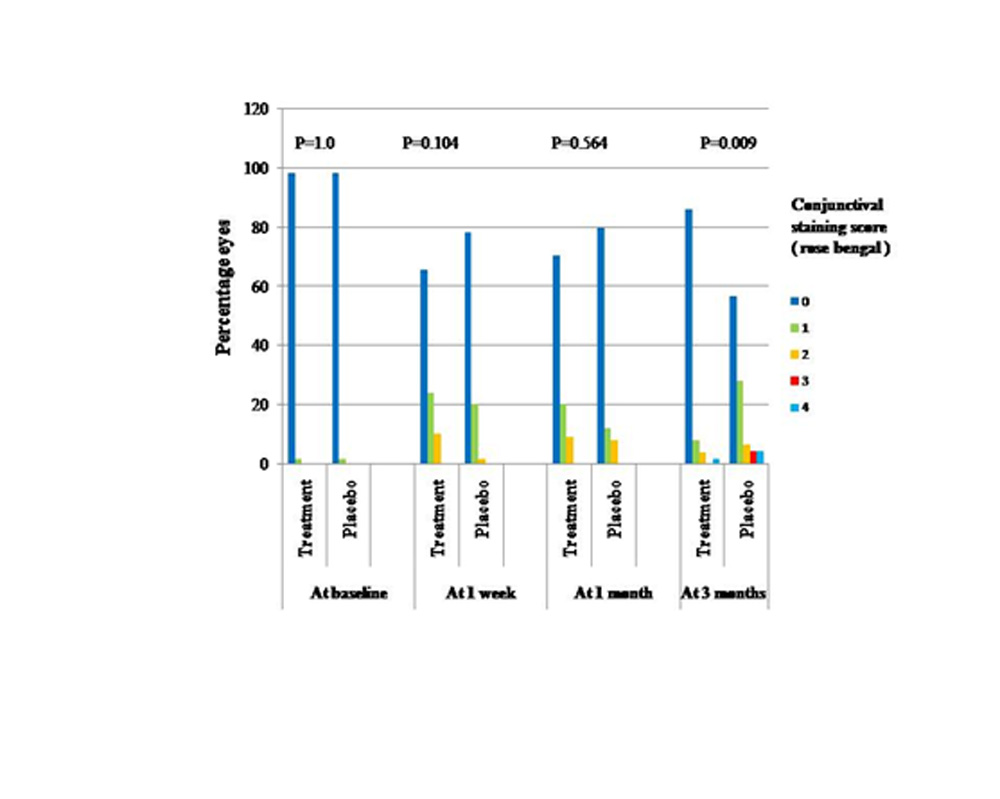
Conjunctival staining scores with Lissamine green dye (Figure2)were similar at baseline with majority (98.3%) of the eyes in both groups having a score of 0, while 1.7% eyes in both groups had a score of 1 (p=1.0). At the last follow up visit i.e. 3 months postoperatively a significantly larger (p=0.009) percentage of patients in the placebo group (43.4%) had some degree of conjunctival staining as compared to the treatment group (14%), though the proportion of eyes having a score of above 3 (which is considered abnormal) was similar in both groups (p=0.606)
Figure 2
Schirmer I without anaesthesia: Table 6shows the mean Schirmer’s readings in the treatment and placebo groups preoperatively and at the subsequent postoperative visits after LASIK. The absolute Schirmer test value was comparable at baseline and at the 1 week and 1 month postoperative visits between the two groups. However at 3 months the Schirmer score in the treatment group (30.52±0.90 mm) was significantly higher than that in the placebo group (25.73 ± 1.26mm) (p=0.003).
| Table 6: Comparison of Schirmer scores Between the Treatment Group and Placebo Group | |||
| Parameter | Treatment Group
Mean ± SEM (millimetres) |
Placebo Group
Mean ± SEM (millimetres) |
P |
| Schirmer Baseline | 25.0 ± 1.17 | 27.49 ± 1.06 | 0.119 |
| Schirmer 1week postop. | 25.55 ± 1.38 | 24.22 ± 1.10 | 0.455 |
| Schirmer 1 month postop. | 26.39 ± 1.28 | 26.80 ± 1.23 | 0.820 |
| Schirmer 3 months postop. | 30.52 ± 0.90 | 25.73 ± 1.26 | 0.003* |
| Post op., postoperative; SEM, standard error of mean; *P<0.05 | |||
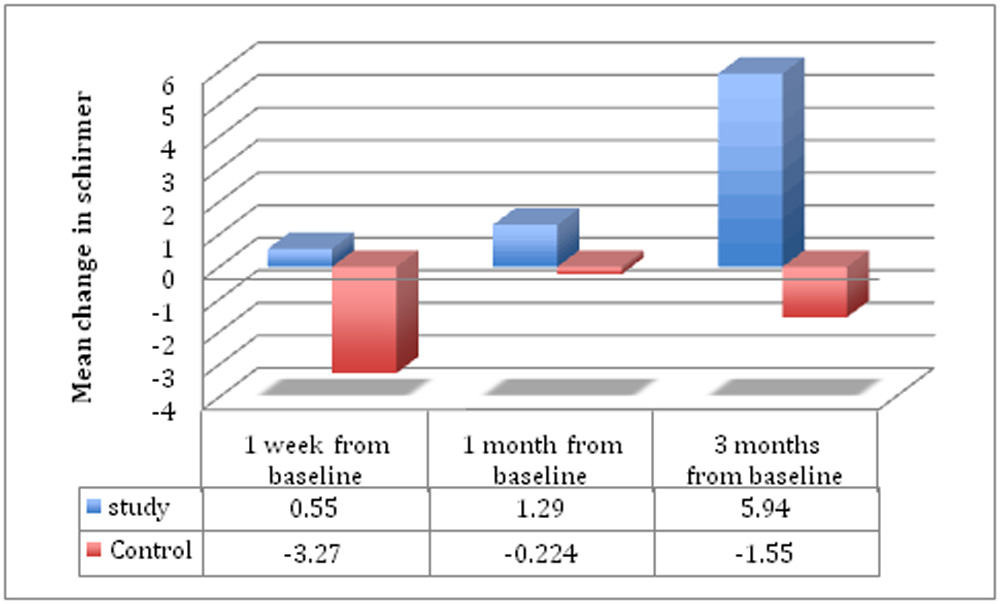
Mean change in Schirmer from baseline was also calculated and within group and inter group comparisons were made (Table 7) .For the treatment group the Schirmer value did not fall below the baseline i.e. preoperative levels at any point during the post operative follow up and was significantly higher as compared to baseline at the 3 month visit (p < 0.01). In comparison the Schirmer score in the placebo group remained depressed below the baseline levels till the end of the 3 months study period.Inter group comparison revealed that the mean change in Schirmer from baseline was significant at 1 week (p=0.016) and at the 3month (p< 0.01) postoperative visits, with the treatment group having better tear secretion (Schirmer) scores at each of these visits.
| Table 7: Inter group comparison of mean change in Schirmer score from baseline | ||||
| Group | Mean ± SEM | Intergroup comparison
(P value) |
||
| Mean change at 1 week postop. from baseline | Treatment | 0.55±1.13 | 0.016* | |
| Placebo | -3.27 ± 1.08 | |||
| Mean change at 1 month postop. from baseline | Treatment | 1.29 ± 1.49 | 0.584 | |
| Placebo | -0.22 ± 1.22 | |||
| Mean change at 3 months postop. from baseline | Treatment | 5.94 ± 1.32 | < 0.01* | |
| Placebo | -1.55 ± 1.23 | |||
| Post op., postoperative; SEM , standard error of mean; *P<0.05
Discussion Omega 3 FA’s have been found to be beneficial in a wide spectrum of dry eye disorders due to multiple mechanisms of action including decreased inflammation and apoptosis of lacrimal gland and ocular surface cells, increased tear secretion and restoration of the lipid layer.29 In the present study which was conducted to determine the effects of oral w3 FA supplementation in preventing signs and symptoms of dry eyes after LASIK a significantly higher (p=0.003) aqueous production was seen in the group receiving w3FA supplementation vis a vis that in the placebo group at 3 months with the power of the study for Schirmer test at 3 months being 93%. Conversely tear film stability as represented by TBUT, though depressed below baseline in both the groups throughout the study period was not significantly better or worse in either the treatment or control groups upto 3 months postoperatively. Previous studies have reported decrease in TBUT 30-32 while Schirmer scores have ranged from no change to moderate depression after LASIK, with the changes persisting for months after the surgery.30,33 NGF concentrations have been shown to increase in tears in the early post-LASIK period.34 Upregulation of NGF production by stromal keratocytes and corneal epithelial cells in response to injury to the cornea has alsobeen documented in literature previously.13 Esquinazi et al 14 and Cortina et al 15 in rabbit models have demonstrated enhanced subepithelial nerve regeneration using DHA in combination with NGF or PEDF. Oral supplementation with w3FA containing DHA in the presence of increased levels of NGF in the post operative period after LASIK, may lead to enhanced nerve regeneration and consequently better restoration of the lacrimal functional unit resulting in an increased tear secretion. This mechanism could be responsible for the better Schirmer scores in the treatment group as compared to the placebo group in our study. Definitive evidence of enhanced corneal nerve regeneration occurring in LASIK patients receiving w 3FA can however only be provided by in vivo confocal microscopy. The anti inflammatory effects of w3FA consumption16-18 may in addition have a beneficial effect on reducing the LASIK induced ocular surface inflammation.10,11 This would result in better functioning of the afferent loop of the lacrimal functional unit, and consequently enhanced secretion of tears from the lacrimal gland. Results of some earlier studies assessing effect of w3FA supplementation on dry eyes secondary to MGD, contact lens use and computer vision syndrome have shown significant improvement in TBUT with no or minimal improvement in Schirmer scores.19,20,24,25 These results appear contradictory to those seen in the present study but may possibly be explained by the difference in the predominant underlying mechanism causing dry eyes in these pathologies vis a vis LASIK associated dry eye. A predominantly evaporative dry eye due to altered lipid layer composition or thickness in MGD patients or those using contact lenses for prolonged durations 35 is more likely to be benefitted from oral w3FA supplementation which help to restore the lipid layer by clearing and thinning the meibomian gland secretions and increasing the w3FA in the polar portion of the tear film’s lipid layer.25,29 This could account for the greater improvement in tear film stability in these populations. Conversely in eyes having undergone LASIK the meibomian gland secretions are not significantly altered. Toda et al3 have documented a drop in the blink rate as being mainly responsible for the increased evaporation in LASIK patients. Blink rate is unlikely to be influenced directly by w-3 FA supplementation and hence tear film stability ( as represented by TBUT) can be expected to be similar in patients receiving w3FA supplementation versus those not receiving it in the initial period after LASIK, as was also the case in the present study. Baseline scores for conjunctival staining were similar in both the groups in the present study , though at the 3 month follow up visit there was trend towards an increased number of patients showing some degree of conjunctival staining with Lissamine green in the placebo group (43.4%), versus that in the treatment group (14%) . Use of the suction ring in LASIK is associated with decreased conjunctival goblet cell density12 and hence decreased mucin production. Previous studies 19,20 using conjunctival impression cytology have reported improvement of goblet cell densities in patients with dry eyes receiving oral w3FA supplementation, and this may account for the lesser number of patients showing conjunctival staining with Lissamine green in the treatment group in our series. OSDI scores reflect the discomfort experienced by patients due to dry eye, with higher scores representing a greater degree of disability. Though OSDI scores remained elevated above the baseline values till the last follow up at 3 months, the scores in both groups were much below 12 ( the cut off value between asymptomatic and mildly symptomatic dry eye26) indicating lack of significant discomfort in the post operative period in either group . This may be due to the fact that patients with pre existing dry eye disease were excluded from the study and also were managed post operatively with topical steroids for 1 month and artificial tears for 3 months in an attempt to maintain normal ocular surface homeostasis.36 This aggressive post operative regimen followed in both the groups may have masked the beneficial effect of w3FA in the treatment group to some degree. The authors would like to acknowledge a few limitations of this study. Since no blinding was done, the possibility of an observation bias exists. However we believe that by the very nature of testing, TBUT is a more subjective parameter than Schirmer scores (which involve reading values of a strip marked specifically for the purpose, hence making recognition of the end point quite definitive). As the main outcome of the study was observation of better tear secretion rates i.e higher Schirmer scores in the treatment group as compared to placebo, while no significant difference was seen in the TBUT, the results in all probability reflect actual outcomes. Other limitations included absence of in vivo confocal microscopy of the anterior corneal nerve complex, lack of measurement of inflammatory tear film markers and a lack of comparison of baseline serum levels of w3FA between the groups. In conclusion, the present study demonstrated that oral supplementation with w3FA in patients undergoing LASIK , had a positive influence on tear secretion ,while tear film stability remained largely unaffected when compared to the placebo group. More studies with a larger patient base and longer follow up durations may help to substantiate the findings of the present series, and determine the role of w3FA supplementation in managing the post LASIK dry eye.
References:
|
||||


Leave a Comment